Online Database of Chemicals from Around the World
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Classification | Organic raw materials >> Organic fluorine compound >> Fluoroaniline series |
|---|---|
| Name | 4,4'-Diamino-2'-chlorobenzanilide |
| Synonyms | 4-Amino-N-(4-amino-2-chlorophenyl)benzamide |
| Molecular Structure | 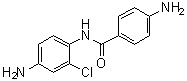 |
| Molecular Formula | C13H12ClN3O |
| Molecular Weight | 261.71 |
| CAS Registry Number | 14108-16-8 |
| SMILES | O=C(C1=CC=C(C=C1)N)NC2=CC=C(C=C2Cl)N |
| Solubility | Practically insoluble (0.029 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.416±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 408.7$+/-$45.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 201.0$+/-$28.7 $degree$C (Calc.)* |
| Index of refraction | 1.736 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details |
| SDS | Available |
|
4,4'-Diamino-2'-chlorobenzanilide is an aromatic diamine compound in which two amino groups are positioned at the 4-positions of a benzene ring system, with a chlorine atom substituted at the 2'-position of the anilide moiety. The structure consists of a benzene ring connected via an amide linkage to a second benzene ring, forming the benzanilide framework. The presence of amino groups provides reactive sites for chemical modification, while the chloro substituent influences electronic properties and steric effects, making the compound useful in polymer synthesis and pharmaceutical chemistry. The molecular structure features an anilide linkage, where the carbonyl group of the amide connects the two aromatic rings. The para-positioned amino groups are highly nucleophilic, allowing reactions such as acylation, sulfonylation, and Schiff base formation. The chlorine atom at the ortho-position of the anilide ring modulates the reactivity of the molecule by exerting an electron-withdrawing effect and introducing steric hindrance, which can influence the selectivity of chemical reactions. Synthesis of 4,4'-diamino-2'-chlorobenzanilide generally involves the condensation of 4,4'-diamino-substituted aromatic amines with 2-chlorobenzoyl chloride under controlled conditions. Protection and deprotection strategies are often employed to prevent side reactions at the amino groups during acylation. Such synthetic procedures yield a high-purity compound suitable for further chemical transformations and material applications. In polymer chemistry, 4,4'-diamino-2'-chlorobenzanilide serves as a monomer for high-performance polymers, including polyamides and polyimides. Its diamine functionality allows the formation of linear or crosslinked polymer chains through reaction with dianhydrides or diacid chlorides. The rigid aromatic backbone, combined with the steric influence of the chloro substituent, imparts thermal stability, mechanical strength, and chemical resistance to the resulting polymers. These materials are used in advanced coatings, adhesives, and electronic components. In addition to polymer applications, the compound is employed as an intermediate in organic synthesis. The amino and amide functionalities allow the preparation of derivatives with tailored properties for medicinal chemistry and materials science. Derivatives can be designed to introduce additional functional groups, modify solubility, or enhance binding interactions for use in drug discovery or as ligands in coordination chemistry. Overall, 4,4'-diamino-2'-chlorobenzanilide is a multifunctional aromatic compound characterized by two reactive amino groups, a benzanilide linkage, and a strategically positioned chloro substituent. Its combination of reactivity, structural rigidity, and electronic modulation makes it an important intermediate for high-performance polymers, organic synthesis, and the development of functional materials. References none |
| Market Analysis Reports |