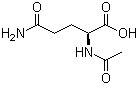
Aceglutamide, also known as N-acetyl-L-glutamine, is a derivative of the naturally occurring amino acid L-glutamine, in which the amide nitrogen of glutamine is acetylated to form an N-acetyl amide bond. Its molecular structure retains the glutamic acid backbone with the acetylated amide side chain, making it more chemically stable than free L-glutamine. Aceglutamide appears as a white to off-white crystalline powder, is slightly soluble in water, and has been used primarily for its potential neurotropic and metabolic effects.
Aceglutamide was developed as a more stable alternative to glutamine for therapeutic applications, particularly in situations where glutamine’s rapid degradation and limited stability posed challenges. L-glutamine is an important amino acid involved in nitrogen transport, immune function, and as a precursor for neurotransmitters such as glutamate and gamma-aminobutyric acid (GABA). However, its direct pharmaceutical use is limited by poor shelf stability. The acetylation of glutamine in aceglutamide enhances its resistance to enzymatic hydrolysis and oxidation, making it more suitable for formulation and administration.
Aceglutamide has been studied and used in various clinical contexts, especially in neurological and cognitive support. It has been investigated for its potential to improve cerebral metabolism, enhance memory and learning, and support recovery from cognitive impairment. Its mechanism of action is not fully established, but it is believed to contribute to increased levels of glutamine and glutamate in the brain, supporting neurotransmitter balance and energy metabolism. Some studies have evaluated its use in age-related cognitive decline and neurodegenerative disorders.
In certain regions, aceglutamide has been incorporated into combination pharmaceutical preparations with other compounds such as vitamin B6, hydroxocobalamin, or nucleotides, intended for use in patients with cerebral vascular disorders, neuroasthenia, or fatigue. These combinations aim to improve neural energy utilization, promote nerve cell repair, or alleviate symptoms associated with central nervous system fatigue or stress.
Aceglutamide has also been explored for parenteral nutrition, particularly in formulations that aim to provide a stable glutamine source for patients undergoing intensive care, surgery, trauma recovery, or cancer therapy. Glutamine is considered conditionally essential in such scenarios, but its instability limits its use. Aceglutamide, due to its greater stability, may serve as a glutamine donor in the body following enzymatic conversion, although its clinical use for this purpose varies by region.
In terms of pharmacokinetics, aceglutamide is absorbed and metabolized to release glutamine in vivo. The exact conversion efficiency and bioavailability depend on the route of administration and physiological conditions. Oral forms have been used in dietary supplements and nootropic formulations, though clinical evidence supporting cognitive enhancement in healthy individuals remains limited.
Aceglutamide has a favorable safety profile at standard therapeutic doses. It is generally well tolerated, with minimal side effects reported in clinical use. However, as with any compound affecting neurotransmitter pathways, caution is advised in patients with seizure disorders or altered mental status. It is not widely approved for use in all countries and may be available only under specific indications or as part of combination therapies in certain regions.
In summary, aceglutamide is a chemically stable derivative of glutamine used for its potential neurotropic and metabolic benefits. It serves as a glutamine donor in formulations aimed at cognitive support, neural recovery, and parenteral nutrition. Its stability and tolerability have made it a useful compound in specialized therapeutic applications, though its broader clinical use is limited and varies internationally.
References
2024. Dysregulation of amino acids balance as potential serum-metabolite biomarkers for diagnosis and prognosis of diabetic retinopathy: a metabolomics study. Journal of diabetes and metabolic disorders, 23(2).
DOI: 10.1007/s40200-024-01462-y
2020. Enhanced thioredoxin, glutathione and Nrf2 antioxidant systems by safflower extract and aceglutamide attenuate cerebral ischaemia/reperfusion injury. Journal of Cellular and Molecular Medicine, 24(10).
DOI: 10.1111/jcmm.15099
|




























































 GHS07 Warning Details
GHS07 Warning Details