Sulfur hexafluoride is a simple inorganic compound composed of one sulfur atom surrounded by six fluorine atoms, with the chemical formula SF6. It is a colorless, odorless, non-flammable, and extremely stable gas under normal conditions. Sulfur hexafluoride was first synthesized in the 1930s through the direct reaction of elemental sulfur with fluorine gas. Its high chemical inertness and unique physical properties have made it a widely used material in industrial, electrical, and medical applications.
The molecular structure of sulfur hexafluoride is octahedral, with six fluorine atoms symmetrically arranged around a central sulfur atom. This geometry results in a highly stable molecule due to the strong S–F covalent bonds and the distribution of electron density that minimizes repulsion between the fluorine atoms. The high bond strength and kinetic inertness prevent SF6 from reacting readily with most chemical reagents, making it chemically inert under a wide range of conditions.
One of the primary industrial applications of sulfur hexafluoride is as an insulating and arc-quenching gas in high-voltage electrical equipment. Its excellent dielectric properties and thermal stability make it ideal for use in circuit breakers, transformers, and gas-insulated switchgear, where it prevents electrical discharge and ensures reliable operation. SF6 is also used in high-voltage testing and as a tracer gas in leak detection due to its inertness and detectability at very low concentrations.
Sulfur hexafluoride is employed in the medical field as a contrast agent in ultrasound imaging. Microbubbles filled with SF6 provide enhanced acoustic reflection, improving the visualization of blood flow and organ structures. Its non-toxic and biologically inert nature makes it safe for temporary introduction into the body for diagnostic purposes.
In scientific research, SF6 is used in particle physics as an electron absorber and as a component in gaseous detectors due to its electronegative properties. It is also utilized in semiconductor manufacturing for plasma etching processes and as a dielectric medium in specialized equipment.
Environmental considerations are important for sulfur hexafluoride. It is a potent greenhouse gas with a global warming potential thousands of times greater than carbon dioxide. Its long atmospheric lifetime, combined with its widespread use in electrical systems, has prompted regulations to minimize emissions and develop recycling or containment strategies. Research is ongoing to find alternative insulating gases with lower environmental impact while maintaining the desirable properties of SF6.
Overall, sulfur hexafluoride is a chemically inert, thermally stable, and highly symmetrical molecule with diverse applications in industry, medicine, and research. Its unique combination of dielectric strength, chemical stability, and safety features has made it an essential material in high-voltage technology and other specialized fields, though its environmental impact requires careful management.
References
2025. Considering sectoral warming and cooling emissions and their lifetimes can improve climate change mitigation policies. npj Climate and Atmospheric Science.
DOI: 10.1038/s41612-025-01131-8
1951. The Coexistence Curve of Sulfur Hexafluoride in the Critical Region. The Journal of physical and colloid chemistry.
DOI: 10.1021/j150487a008
|



























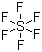

 GHS04;GHS07 Warning Details
GHS04;GHS07 Warning Details