Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Hangzhou StarShine Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.starshinepharm.com | |||
 | +86 (571) 8512-3681 +86 13777804878 | |||
 | +86 (571) 8512-2157 | |||
 | sales@starshinepharm.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shanghai Growingchem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.growingchem.com | |||
 | +86 (21) 5080-0795 | |||
 | +86 (21) 6496-3699 | |||
 | sales@growingchem.com syzhang1983@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Micro-Mega Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.micro-megaindustry.com | |||
 | +86 (21) 5715-3848 3462-8682 | |||
 | +86 (21) 5715-3848 | |||
 | weij@micro-megaindustry.com lewisliu2010@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Boyuan Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.boyuanpharm.com | |||
 | +86 (531) 6995-4981 8896-3280 +86 15806417970 | |||
 | +86 (531) 8896-4879 | |||
 | Jeffrey.Liu@boyuanpharm.com boyuanchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Trignokem International | India | |||
|---|---|---|---|---|
 | www.trignokem.com | |||
 | +91 (40) 2717-0709 | |||
 | info@trignokem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Qingdao Xinnuo Pharmaceutical Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xinnuopharma.com | |||
 | +86 (531) 6656-1050 +86 18615688656 +86 18653156686 | |||
 | +86 (531) 6656-1050 | |||
 | sales@xinnuopharma.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shaoxing Fangxiao Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fangxiaochem.com | |||
 | +86 (571) 2829-7060 +86 18969095308 | |||
 | +86 (571) 2829-7065 | |||
 | sales@fangxiaochem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Enki Biopharmaceuticals (Shanghai) Limited | China | |||
|---|---|---|---|---|
 | www.enkibiopharma.com | |||
 | +86 (21) 5768-0965 +86 13916707528 | |||
 | +86 (21) 5768-0922 | |||
 | info@enkibiopharma.com | |||
 | QQ Chat | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Jinan Lead Pharm-chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leadchem.net | |||
 | +86 13127158727 | |||
 | leadqls@leadchem.net | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| SynChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.synchem.com | |||
 | +1 (847) 298-2436 ex 230 | |||
 | +1 (847) 298-2439 | |||
 | sales@synchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Endeavour Speciality Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.endeavourchem.co.uk | |||
 | +44 (1327) 310-079 | |||
 | +44 (1327) 310-701 | |||
 | enquiries@endeavourchem.co.uk | |||
| Chemical manufacturer | ||||
| Chemcia Scientific, LLC | USA | |||
|---|---|---|---|---|
 | www.chemcia.com | |||
 | +1 (858) 678-0337 | |||
 | sales@chemcia.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Aaron Chemistry GmbH | Germany | |||
|---|---|---|---|---|
 | www.aaron-chemistry.de | |||
 | +49 (8823) 917-521 | |||
 | +49 (8823) 917-523 | |||
 | sales@aaron-chemistry.de | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Amide |
|---|---|
| Name | 4-Hydroxythiobenzamide |
| Synonyms | 4-Hydroxybenzene-1-carbothioamide |
| Molecular Structure | 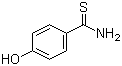 |
| Molecular Formula | C7H7NOS |
| Molecular Weight | 153.20 |
| CAS Registry Number | 25984-63-8 |
| EC Number | 674-712-0 |
| SMILES | C1=CC(=CC=C1C(=S)N)O |
| Melting point | 181-185 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H317-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P272-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
4-Hydroxythiobenzamide (4-HTB) was discovered through efforts to develop thiobenzamide derivatives with enhanced biological activity and therapeutic potential. This compound, characterized by a hydroxy group and a thiobenzamide structure, was synthesized via modification of benzamide compounds to introduce sulfur-containing functional groups. The discovery process involved classical organic synthesis techniques, focusing on the strategic placement of the hydroxy group on the benzene ring to explore its effects on biological activity. Researchers aimed to utilize the unique properties of sulfur-containing compounds in drug design, leading to the creation of 4-HTB as a promising molecule for further study. 4-HTB exhibits promising antibacterial properties against a range of Gram-positive and Gram-negative bacteria. The presence of the hydroxy and thio groups enhances its ability to disrupt bacterial cell membranes and interfere with essential bacterial enzymes. This makes 4-HTB a potential candidate for developing new antibacterial agents to combat resistant bacterial strains, addressing the growing issue of antibiotic resistance. Besides antibacterial effects, 4-HTB has shown antifungal activity against several pathogenic fungi. It can inhibit fungal growth by disrupting cell wall synthesis and membrane integrity. This dual antibacterial and antifungal activity broadens its potential use in treating various infectious diseases, making it a valuable addition to antifungal therapy development. 4-HTB acts as a potent antioxidant, scavenging free radicals and reducing oxidative stress. Its antioxidant activity is attributed to the hydroxy group, which can donate electrons to neutralize reactive oxygen species (ROS). This property is valuable in developing therapeutic agents to protect against oxidative stress-related diseases, such as neurodegenerative disorders and cardiovascular diseases. In addition to antioxidant effects, 4-HTB exhibits anti-inflammatory properties by modulating inflammatory pathways. It can inhibit pro-inflammatory cytokines and reduce inflammation in various models of inflammatory diseases. This makes 4-HTB a potential candidate for developing anti-inflammatory drugs to treat conditions like arthritis and inflammatory bowel disease. 4-HTB serves as a valuable lead compound in drug discovery programs aimed at developing new therapeutic agents. Its unique chemical structure and biological activities make it a versatile scaffold for designing derivatives with enhanced efficacy and selectivity. Researchers can modify the hydroxy and thio groups to create new analogs with improved pharmacological profiles, leading to potential new treatments for various diseases. Emerging studies suggest that 4-HTB may have anticancer properties. It can induce apoptosis (programmed cell death) and inhibit the proliferation of cancer cells in certain types of cancer. Its ability to modulate key signaling pathways involved in cell survival and apoptosis makes it a promising candidate for further research in cancer therapy. 4-HTB can be used as a biochemical probe to study the roles of thiobenzamide derivatives in biological systems. Its interactions with biomolecules and cellular components provide insights into the mechanisms of action of similar compounds, aiding in the design of new biochemical tools and therapeutic agents. In analytical chemistry, 4-HTB can serve as a standard for developing and validating analytical methods to detect and quantify thiobenzamide derivatives in various matrices. This is valuable for quality control and regulatory compliance in pharmaceutical and chemical industries. References 2009. P-Alkylation of Dihalophosphines. Science of Synthesis. URL: SD-042-00205 2009. From Alkyldihalophosphines and Organometallic Alkali Metal Compounds or Grignard Reagents. Science of Synthesis. URL: SD-042-00087 2009. Reduction of Halophosphines with Lithium Aluminum Hydride or Other Metal Hydrides. Science of Synthesis. URL: SD-042-00093 |
| Market Analysis Reports |