Online Database of Chemicals from Around the World
| Alway Chem, China | China | |||
|---|---|---|---|---|
 | www.alwaychem.net | |||
 | +86 (532) 8586-4000 | |||
 | +86 (532) 8589-3133 | |||
 | alwaychem@gmail.com alwaychem@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Dafeng Chemical Com.,Ltd. | China | |||
|---|---|---|---|---|
 | www.qzdafeng.cn | |||
 | +86 16773308882 | |||
 | 927608786@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Natural product >> Natural phenols |
|---|---|
| Name | 2-Amino-5-chlorophenol |
| Synonyms | 4-Chloro-2-hydroxyaniline |
| Molecular Structure | 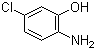 |
| Molecular Formula | C6H6ClNO |
| Molecular Weight | 143.57 |
| CAS Registry Number | 28443-50-7 |
| EC Number | 249-020-3 |
| SMILES | C1=CC(=C(C=C1Cl)O)N |
| Density | 1.4$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 145 - 153 $degree$C (Expl.) |
| Boiling point | 267.7$+/-$25.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 115.7$+/-$23.2 $degree$C (Calc.)* |
| Index of refraction | 1.651 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
2-Amino-5-chlorophenol is an aromatic organic compound containing amino, hydroxyl, and chloro substituents attached to a benzene ring. Its molecular formula is C6H6ClNO. The compound belongs to the class of aminophenols, which are important intermediates in organic synthesis, dye chemistry, and pharmaceutical chemistry. Structurally, the molecule consists of a phenol ring bearing a hydroxyl group (–OH), an amino group (–NH2), and a chlorine atom (–Cl). In the standard numbering system for phenols, the hydroxyl group occupies position 1, the amino group is located at position 2 (ortho to the hydroxyl group), and the chlorine substituent is located at position 5. This substitution pattern creates a multifunctional aromatic system with both electron-donating and electron-withdrawing substituents. The hydroxyl and amino groups are strongly electron-donating through resonance interactions, increasing electron density on the aromatic ring and influencing reactivity toward electrophilic substitution. The chlorine atom exerts an electron-withdrawing inductive effect while also participating weakly in resonance donation. The combined effects of these substituents determine the regioselectivity and chemical behavior of the molecule. The ortho relationship between the amino and hydroxyl groups allows intramolecular hydrogen bonding, which can influence molecular conformation, melting behavior, and solubility. Both functional groups are chemically reactive: the amino group can participate in diazotization, acylation, alkylation, and condensation reactions, while the phenolic hydroxyl group can undergo etherification, esterification, and oxidation reactions. 2-Amino-5-chlorophenol is widely used as an intermediate in the synthesis of dyes, pigments, pharmaceuticals, and agrochemicals. Aminophenol derivatives are particularly important in azo dye chemistry because the amino group can be converted into diazonium salts, which then undergo azo coupling reactions to form colored aromatic azo compounds. In pharmaceutical and fine chemical synthesis, chlorinated aminophenols serve as versatile intermediates for constructing more complex heterocyclic and aromatic compounds. The chlorine substituent can participate in nucleophilic aromatic substitution reactions under appropriate conditions, enabling further functionalization of the aromatic ring. The compound can be synthesized through several routes involving nitration, chlorination, reduction, or hydroxylation reactions on substituted aromatic precursors. One common strategy involves chlorination of aminophenol derivatives or reduction of corresponding nitrochlorophenols. From a physicochemical perspective, 2-amino-5-chlorophenol exhibits both acidic and basic properties because of the phenolic hydroxyl and amino groups. The hydroxyl group can lose a proton under strongly basic conditions, while the amino group can be protonated in acidic media. This amphoteric behavior affects solubility and reactivity in aqueous systems. The aromatic ring and conjugated substituents also contribute to characteristic ultraviolet absorption behavior. Oxidation of aminophenols can produce quinone-imine or related oxidation products, which is important in both synthetic chemistry and stability considerations. Historically, substituted aminophenols became important industrial intermediates during the expansion of synthetic dye manufacturing in the late 19th and early 20th centuries. Their versatile reactivity and ability to form intensely colored azo compounds made them valuable components in industrial organic chemistry. Overall, 2-amino-5-chlorophenol is a multifunctional chlorinated aminophenol containing amino, hydroxyl, and chloro substituents on an aromatic ring. Its combination of nucleophilic, acidic, and electrophilic reactivity makes it an important intermediate in dye synthesis, pharmaceutical chemistry, and aromatic functionalization reactions. References 2025. Exploring Schiff Bases Derived from 2-hydroxybenzaldehyde as Potential Anticancer Agents: Synthesis, Characterization, Molecular Docking and in-vitro Evaluation. Cell Biochemistry and Biophysics. DOI: 10.1007/s12013-025-01826-y |
| Market Analysis Reports |