Online Database of Chemicals from Around the World
| Jiangxi Kingnord Industrial Limited | China | |||
|---|---|---|---|---|
 | www.kingnordchem.com | |||
 | +86 (791) 8649-3591 | |||
 | +86 (791) 8649-3642 | |||
 | jeff@kingnord.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Sigma-Aldrich, Inc. | China | |||
|---|---|---|---|---|
 | www.sigmaaldrich.com | |||
 | +86 (21) 6141-5566 800-819-3336 | |||
 | orderCN@sial.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2013 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Amide |
|---|---|
| Name | N,N'-Hexamethylenebisacetamide |
| Synonyms | Hexamethylenediacetamide; N,N'-Diacetylhexamethylenediamine; 1,6-Hexanebisacetamide |
| Molecular Structure | 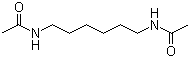 |
| Molecular Formula | C10H20N2O2 |
| Molecular Weight | 200.28 |
| CAS Registry Number | 3073-59-4 |
| SMILES | CC(=O)NCCCCCCNC(=O)C |
| Density | 1.0$+/-$0.1 g/cm3 Calc.*, 0.974 g/mL (Expl.) |
|---|---|
| Melting point | 128 - 129 $degree$C (Expl.) |
| Boiling point | 456.7$+/-$28.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 193.2$+/-$24.2 $degree$C (Calc.)* |
| Index of refraction | 1.454 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
N,N′-Hexamethylenebisacetamide is a symmetric organic compound belonging to the class of bis-amides. It consists of a hexamethylene (six-carbon) aliphatic chain linking two acetamide groups through nitrogen atoms. Its molecular formula is C10H20N2O2. Structurally, the molecule can be described as two acetamide units (CH3–C(=O)–NH–) connected via their nitrogen atoms to opposite ends of a linear hexamethylene spacer (–(CH2)6–). This gives the compound a flexible, extended chain structure with two terminal amide functionalities. The amide bonds introduce polarity and hydrogen-bonding capability, while the hydrocarbon chain contributes hydrophobic character. The amide functional groups are stabilized by resonance between the carbonyl oxygen and the nitrogen lone pair, resulting in partial double-bond character of the C–N bonds. This resonance restricts rotation around the amide linkage and contributes to the overall conformational properties of the molecule. The compound can participate in hydrogen bonding as both donor (N–H) and acceptor (C=O), which influences its physical behavior. N,N′-Hexamethylenebisacetamide is typically synthesized through acylation reactions involving hexamethylenediamine and acetylating agents such as acetic anhydride or acetyl chloride. In this process, both amine groups are converted into acetamide functionalities, yielding the symmetrical bisamide product. From a physicochemical perspective, the compound exhibits intermediate polarity due to the presence of two amide groups balanced by a hydrophobic aliphatic chain. This dual character affects its solubility, allowing limited interaction with both polar and nonpolar solvents depending on conditions. Hydrogen bonding between molecules can also influence its melting behavior and solid-state structure. Bis-amides such as N,N′-hexamethylenebisacetamide are of interest in polymer chemistry and materials science because of their ability to form hydrogen-bonded networks. The presence of two amide groups in a flexible chain allows them to act as building blocks or additives in supramolecular assemblies, polyamide-related systems, and coordination complexes. The compound can also serve as an intermediate in organic synthesis, particularly in the preparation of more complex nitrogen-containing derivatives. The amide groups can undergo hydrolysis under acidic or basic conditions to regenerate amines and carboxylic acids, or participate in further functionalization reactions under appropriate conditions. Historically, aliphatic bisamides have been studied in relation to hydrogen bonding behavior, polymer precursor chemistry, and molecular conformational analysis. Their structural simplicity combined with strong intermolecular interactions makes them useful model compounds in physical organic chemistry. Overall, N,N′-hexamethylenebisacetamide is a symmetrical aliphatic bisamide composed of two acetamide groups linked by a flexible six-carbon chain. Its combination of hydrogen-bonding functionality and hydrocarbon flexibility gives it properties relevant to materials chemistry, supramolecular interactions, and synthetic organic chemistry. References 2026. World free HIV: the novel therapeutic approaches for eliminating latent HIV infection. Virology Journal. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12933922 2025. Histone lysine demethylases in breast cancer: molecular mechanisms, biological functions, and therapeutic intervention. Molecular Cancer. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12882162 |
| Market Analysis Reports |