Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Tianjin Jinhui Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jinhuipharm.com | |||
 | +86 (22) 2793-2148 2739-0972 | |||
 | +86 (22) 2793-1803 | |||
 | jinhui@jinhuipharm.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhejiang Xianju Xianle Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xianle.com | |||
 | +86 (576) 8778-9188 | |||
 | +86 (576) 8779-3388 | |||
 | zhujian@xianle.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Bondchemistry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bondchemistry.com | |||
 | +86 15858185835 | |||
 | sales@bondchemistry.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2020 | ||||
| Dalton Pharma Services | Canada | |||
|---|---|---|---|---|
 | www.dalton.com | |||
 | +1 (416) 661-2102 | |||
 | +1 (416) 661-2108 | |||
 | chemist@dalton.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | 3,20-Dioxopregna-1,4,9(11),16-tetraen-21-yl acetate |
| Synonyms | 21-Hydroxypregna-1,4,9(11),16-tetraene-3,20-dione 21-acetate |
| Molecular Structure | 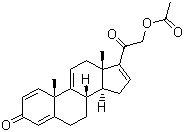 |
| Molecular Formula | C23H26O4 |
| Molecular Weight | 366.46 |
| CAS Registry Number | 37413-91-5 |
| EC Number | 253-497-3 |
| SMILES | CC(=O)OCC(=O)C1=CC[C@@H]2[C@@]1(CC=C3[C@H]2CCC4=CC(=O)C=C[C@@]43C)C |
| Hazard Symbols |    GHS07;GHS08;GHS09 Warning Details GHS07;GHS08;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335-H361-H400 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P318-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
3,20-Dioxopregnan-1,4,9(11),16-tetraen-21-yl acetate, also known as dexamethasone acetate, is a synthetic glucocorticoid derivative widely used in medicine and pharmacology for its potent anti-inflammatory and immunosuppressive properties. Dexamethasone acetate belongs to the corticosteroid family and was first synthesized in the mid-20th century as an analog of natural corticosteroids. Its synthesis involves the acetylation of dexamethasone, a well-known glucocorticoid, at the 21-hydroxyl group. This modification enhances its pharmacokinetic properties, particularly its lipid solubility and metabolic stability, thereby optimizing its therapeutic efficacy. Chemically, dexamethasone acetate is characterized by a pregnane structure with double bonds at positions 1 and 4, a ketone at positions 3 and 20, and an acetate group at position 21. This molecular structure confers high potency and a longer duration of action compared to natural glucocorticoids. It is usually formulated as a white crystalline powder or an injectable solution for clinical use. Dexamethasone acetate exerts a powerful anti-inflammatory effect by inhibiting the synthesis of inflammatory mediators such as prostaglandins and leukotrienes. It suppresses the immune response by reducing cytokines and chemokines involved in inflammatory pathways, making it effective in treating conditions such as arthritis, asthma, and dermatitis. As an immunosuppressant, dexamethasone acetate inhibits the activation and proliferation of immune cells. It is used to prevent transplant rejection and treat autoimmune diseases in which the immune system attacks healthy tissue, such as systemic lupus erythematosus (SLE) and rheumatoid arthritis. Dexamethasone acetate can be taken orally, topically, or injected, depending on the condition being treated. It is indicated for a variety of medical conditions, including allergic reactions, respiratory diseases (e.g., COPD), and certain cancers (e.g., leukemia and lymphoma), where its anti-inflammatory and immunosuppressive effects are effective. Clinical uses include treatment of adrenal insufficiency (e.g., Addison's disease) and pituitary disorders that affect corticosteroid production. Treatment of ocular inflammation (e.g., uveitis, allergic conjunctivitis) and prevention of inflammation following ocular surgery. Topical preparations are used to relieve itching, inflammation, and allergic skin reactions. While dexamethasone acetate is effective in treating a variety of conditions, long-term use or high doses may result in adverse effects such as osteoporosis, hyperglycemia, and increased susceptibility to infection. Careful monitoring and dose adjustment are essential to minimize these risks. References 2019. Synthesis of 13C-Labeled Steroids. Synthesis, 51(18). DOI: 10.1055/s-0037-1611914 2018. Efficient Synthesis of 21-Acetoxypregna-1,4,9(11),16-Tetraene-3,20-Dione, a Key Intermediate in the Synthesis of Highly Active Fluorinated Corticosteroids from 9α-Hydroxyandrostenedione. Pharmaceutical Chemistry Journal, 52(8). DOI: 10.1007/s11094-018-1898-y 2017. Steroid Bioconversions. Methods in Molecular Biology, 1645. DOI: 10.1007/978-1-4939-7183-1_1 |
| Market Analysis Reports |