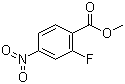
Methyl 2-fluoro-4-nitrobenzoate is an organic compound with the chemical formula C8H6FNO4. It consists of a benzoate ester functional group attached to a benzene ring, which is substituted with a fluoro group at the 2-position and a nitro group at the 4-position. This compound is part of a larger class of substituted benzoates, which are often used in various chemical and industrial applications. Methyl 2-fluoro-4-nitrobenzoate is synthesized through straightforward organic reactions and has garnered attention due to its reactivity and potential applications in several fields, including pharmaceuticals, agrochemicals, and materials science.
The discovery of methyl 2-fluoro-4-nitrobenzoate is part of ongoing research into functionalized aromatic compounds that possess useful electronic properties due to the presence of electron-withdrawing groups such as the fluoro and nitro groups. These types of substitutions can significantly alter the chemical behavior and reactivity of the benzene ring, making compounds like methyl 2-fluoro-4-nitrobenzoate useful for a variety of synthetic applications. The synthesis of this compound typically involves nitration of methyl 2-fluorobenzoate, followed by esterification to yield the final product. The introduction of both the nitro and fluoro groups can be done efficiently under controlled conditions using standard organic synthesis techniques.
Methyl 2-fluoro-4-nitrobenzoate has a variety of applications, particularly in the pharmaceutical industry. The electron-withdrawing properties of the nitro and fluoro groups can influence the biological activity of compounds derived from this molecule. Fluoro-substituted aromatic compounds are often found to exhibit enhanced metabolic stability and bioavailability, which makes them valuable building blocks in drug discovery and development. Methyl 2-fluoro-4-nitrobenzoate is used in the synthesis of various drug-like molecules that target different biological pathways. The presence of the nitro group adds an additional layer of reactivity, making the compound suitable for further functionalization and modification into more complex bioactive molecules.
Another notable application of methyl 2-fluoro-4-nitrobenzoate lies in the field of agrochemicals. Benzoate esters with fluoro and nitro substitutions are often explored as potential pesticides, herbicides, or fungicides.
References
2017. Venetoclax. Pharmaceutical Substances.
URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-22-0046
|
































 GHS07 Warning Details
GHS07 Warning Details