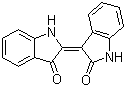
Indirubin is a naturally occurring bis-indole alkaloid primarily isolated from Indigofera tinctoriaand other indigo-producing plants. It is one of the active components of traditional Chinese medicine formulations, particularly in the treatment of chronic myelogenous leukemia and other hematological disorders. Structurally, indirubin is an isomer of indigo, characterized by its two indole moieties connected via a double-bonded carbonyl system, which confers its unique biological activities.
The discovery of indirubin dates back to the 19th century, when it was first identified as a pigment responsible for the red coloration in some indigo extracts. Subsequent investigations revealed that indirubin exhibits significant pharmacological properties distinct from its parent indigo compound. Early studies in traditional medicine highlighted its effectiveness in treating blood disorders, and modern research has focused on elucidating its molecular mechanisms and potential therapeutic applications.
Pharmacologically, indirubin is recognized for its antiproliferative and antitumor activities. It functions as an inhibitor of cyclin-dependent kinases (CDKs) and glycogen synthase kinase-3 (GSK-3), which play critical roles in cell cycle regulation and signaling pathways associated with tumor growth. By modulating these targets, indirubin can induce cell cycle arrest and apoptosis in malignant cells, making it a promising candidate for anticancer therapy. Research has also demonstrated its potential efficacy against leukemia, glioma, and other cancer types.
In addition to its anticancer effects, indirubin exhibits anti-inflammatory and neuroprotective activities. It can suppress the production of pro-inflammatory cytokines and reduce oxidative stress, contributing to its therapeutic potential in inflammatory and neurodegenerative conditions. These properties are mediated through the modulation of signaling pathways such as NF-κB, JAK/STAT, and MAPK, which are involved in inflammation and cellular stress responses.
The bioavailability of indirubin is relatively limited due to its poor water solubility and rapid metabolism, which has led to the development of various formulations to enhance its pharmacokinetic properties. Techniques such as nanoparticle encapsulation, liposomal delivery, and chemical modifications have been employed to improve solubility, stability, and tissue distribution, thereby increasing its therapeutic efficacy in preclinical models.
Clinically, indirubin has been used in traditional Chinese medicine for the management of leukemia and other chronic blood disorders, often as part of multi-component herbal formulations. Contemporary research continues to explore its potential as a lead compound for anticancer drug development, with a focus on understanding its molecular targets, optimizing its pharmacological profile, and evaluating its safety in clinical settings.
Overall, indirubin is a bioactive bis-indole alkaloid with a long history of medicinal use and demonstrated pharmacological activities. Its antiproliferative, anti-inflammatory, and neuroprotective effects, combined with ongoing advances in formulation and delivery strategies, highlight its potential for development as a therapeutic agent in oncology and other disease areas.
References
2025. Synergistic Sedative Activity of Indirubin on Diazepam in Thiopental Sodium-induced Sleeping Mice: A Potential GABAergic Transmission. Molecular Neurobiology.
DOI: 10.1007/s12035-025-05176-x
2008. Photoreactivity of indirubin derivatives. Photochemical & Photobiological Sciences, 7(3).
DOI: 10.1039/b711261k
1951. Effect of indirubin on white blood cell count of the guinea pig. Cellular and Molecular Life Sciences, 7(8).
DOI: 10.1007/bf02150557
|



























































 GHS07 Warning Details
GHS07 Warning Details