Online Database of Chemicals from Around the World
| Shanghai SPE Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.spechemicals.com | |||
 | +86 (21) 5138-6314 | |||
 | +86 (21) 5138-6414 | |||
 | info@spechemicals.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Koffee Pharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.koffeepharm.com | |||
 | +86 (21) 6172-9394 | |||
 | +86 (21) 6172-9329 | |||
 | sales01@koffeepharm.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Wuhan Roche Technology Development Co., Ltd | China | |||
|---|---|---|---|---|
 | www.luoshikg.com | |||
 | +86 18986168071 | |||
 | +86 (027) 8812-1016 | |||
 | 18986168071@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Archimica Inc. | USA | |||
|---|---|---|---|---|
 | www.archimica.com | |||
 | +1 (302) 994-3043 | |||
 | +1 (302) 994-3475 | |||
 | contact@archimica.com | |||
| Chemical manufacturer since 1946 | ||||
| OChem Incorporation | USA | |||
|---|---|---|---|---|
 | www.ocheminc.com | |||
 | +1 (847) 403-7044 | |||
 | +1 (847) 298-5008 | |||
 | sales@ocheminc.com | |||
| Chemical manufacturer | ||||
| Anichem LLC | USA | |||
|---|---|---|---|---|
 | www.anichemllc.com | |||
 | +1 (732) 821-6500 | |||
 | +1 (732) 821-6008 | |||
 | sales@anichemllc.com | |||
| Chemical manufacturer | ||||
| Euticals S.p.A. | Italy | |||
|---|---|---|---|---|
 | www.euticals.com | |||
 | +39 (371) 490-2339 | |||
 | +39 (371) 417-635 | |||
 | contact@archimica.com | |||
| Chemical manufacturer since 1946 | ||||
| Alchymars SpA | Italy | |||
|---|---|---|---|---|
 | www.trifarma.it | |||
 | +39 (2) 969-0018 | |||
 | +39 (2) 969-0643 | |||
 | trifarma@trifarma.it | |||
| Chemical manufacturer | ||||
| SynInnova Laboratories Inc. | USA | |||
|---|---|---|---|---|
 | www.syninnova.com | |||
 | +1 (780) 485-2962 | |||
 | +1 (780) 485-2963 | |||
 | sales@syninnova.com | |||
| Chemical manufacturer | ||||
| Hunan Steroid Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.steroidchem.com | |||
 | +86 (731) 8276-2659 8276-2868 8276-2898 | |||
 | +86 (731) 8555-1702 | |||
 | info@steroidchem.com sun@steroidchem.com | |||
| Chemical manufacturer since 2002 | ||||
| Classification | API >> Hormone and endocrine-regulating drugs >> Androgen and anabolic hormone drugs |
|---|---|
| Name | Oxandrolone |
| Synonyms | 17beta-Hydroxy-17-methyl-2-oxa-5alpha-androstan-3-one |
| Molecular Structure | 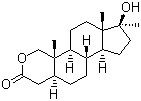 |
| Molecular Formula | C19H30O3 |
| Molecular Weight | 306.44 |
| CAS Registry Number | 53-39-4 |
| EC Number | 200-172-9 |
| SMILES | C[C@]12CC[C@H]3[C@H]([C@@H]1CC[C@]2(C)O)CC[C@@H]4[C@@]3(COC(=O)C4)C |
| Solubility | 41 mg/mL (CHCl3) (Expl.) |
|---|---|
| Density | 1.1±0.1 g/cm3, Calc.* |
| Melting point | 235-238 °C |
| Index of Refraction | 1.526, Calc.* |
| Boiling Point | 444.4±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 179.1±21.5 °C, Calc.* |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H312-H332-H351-H360 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P271-P280-P302+P352-P304+P340-P317-P318-P321-P362+P364-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| Controlled Substance | DEA Drug Code Number: 4000 Details | ||||||||||||||||||||||||||||||||||||||||
| CSA Schedule: III | |||||||||||||||||||||||||||||||||||||||||
| Narcotics? No | |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Oxandrolone is a synthetic anabolic steroid first developed in 1962 by Searle Laboratories. It was initially designed to promote weight gain in patients suffering from severe weight loss due to chronic infections, surgery, or prolonged corticosteroid use. The discovery of oxandrolone provided a therapeutic option with a favorable anabolic-to-androgenic ratio, making it well-suited for medical applications while minimizing androgenic side effects. The synthesis of oxandrolone involves the modification of dihydrotestosterone (DHT), replacing the second carbon atom with an oxygen group. This alteration enhances its anabolic properties while reducing its interaction with androgen receptors, contributing to its relatively mild effects on secondary sexual characteristics. Due to its structural modifications, oxandrolone exhibits lower hepatotoxicity compared to other 17α-alkylated anabolic steroids. Oxandrolone is widely used in clinical settings for muscle-wasting conditions, severe burns, osteoporosis, and Turner syndrome. Its ability to stimulate protein synthesis and muscle regeneration has also led to its use in post-surgical recovery and chronic disease management. Additionally, oxandrolone has been explored as a potential treatment for cachexia associated with HIV/AIDS and cancer, demonstrating effectiveness in improving body composition and overall patient well-being. Beyond medical applications, oxandrolone has been misused in athletic and bodybuilding communities due to its muscle-enhancing effects and low androgenic profile. This non-medical use has led to regulatory restrictions in many countries. Despite concerns over misuse, oxandrolone remains a valuable pharmaceutical compound due to its safety profile and therapeutic benefits in controlled medical settings. The continued research into oxandrolone’s mechanisms and potential applications highlights its role in modern medicine. Advances in steroid chemistry and endocrinology may further refine its usage and therapeutic scope in the future. References 2011. Burns: where are we standing with propranolol, oxandrolone, recombinant human growth hormone, and the new incretin analogs? Current Opinion in Clinical Nutrition and Metabolic Care, 14(2). DOI: 10.1097/mco.0b013e3283428df1 2021. Can conditions of skeletal muscle loss be improved by combining exercise with anabolic-androgenic steroids? A systematic review and meta-analysis of testosterone-based interventions. Reviews in Endocrine and Metabolic Disorders, 22(4). DOI: 10.1007/s11154-021-09634-4 1976. Lack of enhanced microsomal enzyme activity by oxandrolone, an inducer of hepatic smooth endoplasmic reticulum. Biochemical Pharmacology, 25(13). DOI: 10.1016/0006-2952(76)90077-0 |
| Market Analysis Reports |