Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Jiangsu Kangheng Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kanghengchem.com | |||
 | +86 (513) 8768-6609 | |||
 | +86 (513) 8768-6600 | |||
 | zkpeasy@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Nanjing Lianye Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lianyechem.com | |||
 | +86 (25) 8358-5062 | |||
 | sales@lianyechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Tetrahedron Scientific Inc. | China | |||
|---|---|---|---|---|
 | www.thsci.com | |||
 | +86 (571) 8848-9473 +86 18072878644 | |||
 | +86 (571) 8848-9473 | |||
 | sales@thsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Nanjing Ningkang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ningkangchem.com | |||
 | +86 (25) 8530-7523 | |||
 | +86 (25) 8549-3049 | |||
 | yxg@longyanchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Honovo Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.honovochem.com | |||
 | +86 (21) 5892-6067 | |||
 | +86 (21) 5892-6067 | |||
 | sales@honovochem.com | |||
| Chemical distributor since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Kessie Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kessiechem.com | |||
 | +86 (519) 8698-7916 +86 15851999766 | |||
 | +86 (519) 8690-8212 | |||
 | info@kessiechem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2018 | ||||
| EnBridge PharmTech (Wuxi) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enbridgepharm.com | |||
 | +86 (510) 8359-1909 +86 18018392058 | |||
 | +86 (510) 8359-1909 | |||
 | info@enbridgepharm.com | |||
 | QQ Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Nanjing Hike Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njhkchem.com | |||
 | +86 (25) 5211-0956 | |||
 | +86 (25) 5211-0956 | |||
 | sophia@njhkchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 17372769668 | |||
 | WhatsApp:17372769668 | |||
| Chemical distributor since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Xi'an Huayao Pharm-Chem Institute | China | |||
|---|---|---|---|---|
 | www.huayaochem.com | |||
 | +86 (29) 8624-6986 | |||
 | +86 (29) 8626-5661 | |||
 | info@huayaochem.com | |||
| Chemical manufacturer since 1999 | ||||
| Aaron Chemistry GmbH | Germany | |||
|---|---|---|---|---|
 | www.aaron-chemistry.de | |||
 | +49 (8823) 917-521 | |||
 | +49 (8823) 917-523 | |||
 | sales@aaron-chemistry.de | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Trigona oHG | Germany | |||
|---|---|---|---|---|
 | www.trigona.de | |||
 | +49 (611) 962-5283 | |||
 | +49 (611) 962-9032 | |||
 | info@trigona.de | |||
| Chemical manufacturer | ||||
| Maybridge Chemical Co., Ltd. | Belgium | |||
|---|---|---|---|---|
 | www.maybridge.com | |||
 | +32 (14) 593-434 | |||
 | maybridge.sales@thermofisher.com | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic aldehyde (containing acetal, hemiacetal) |
|---|---|
| Name | 2-Nitrobenzaldehyde |
| Synonyms | o-Nitrobenzaldehyde |
| Molecular Structure | 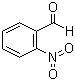 |
| Molecular Formula | C7H5NO3 |
| Molecular Weight | 151.12 |
| CAS Registry Number | 552-89-6 |
| EC Number | 209-025-3 |
| SMILES | C1=CC=C(C(=C1)C=O)[N+](=O)[O-] |
| Melting point | 41-44 °C |
|---|---|
| Boiling point | 153 °C (23 torr) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335-H412 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P273-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P312-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
2-Nitrobenzaldehyde was first synthesized in the late 19th century as part of the broader exploration of aromatic aldehydes and nitro compounds. This compound is characterized by a nitro group (–NO2) positioned ortho to the aldehyde group (–CHO) on a benzene ring. The synthesis of 2-nitrobenzaldehyde typically involves nitration of benzaldehyde or similar precursors under controlled conditions. Its discovery and subsequent availability provided chemists with a versatile intermediate for various organic reactions and applications. 2-Nitrobenzaldehyde serves as a valuable intermediate in organic synthesis. It is used in the preparation of a variety of complex molecules, including heterocycles, which are important in medicinal chemistry. Its reactive aldehyde group can participate in various condensation reactions, such as the Knoevenagel condensation, to form compounds that are useful in further synthetic transformations. The presence of the nitro group also allows for subsequent reduction to form amino derivatives, expanding its utility in multi-step synthesis protocols. In the pharmaceutical industry, 2-nitrobenzaldehyde is employed in the synthesis of active pharmaceutical ingredients (APIs) and intermediates. It is used to create compounds that exhibit biological activity, including anti-inflammatory, antimicrobial, and anticancer properties. The nitro group can be reduced to an amine, which is a common motif in many drugs. This versatility makes 2-nitrobenzaldehyde a crucial building block in drug development and medicinal chemistry. 2-Nitrobenzaldehyde is used in the development of photoresponsive materials. Its ability to undergo photochemical reactions, such as photoinduced isomerization and decomposition, makes it useful in designing light-sensitive compounds and materials. These materials have applications in areas such as optical data storage, photolithography, and the development of smart materials that change properties in response to light exposure. The compound is also a precursor in the synthesis of dyes and pigments. The nitro group can be transformed into various functional groups that impart color to the dye molecules. These dyes are used in textile industries, inks, and coatings, where specific hues and properties are required. In biological research, 2-nitrobenzaldehyde is utilized as a reagent for probing the mechanisms of enzyme activity and protein function. It can be used to modify amino acid residues in proteins, helping researchers study enzyme active sites and protein-ligand interactions. Its use in these studies aids in understanding fundamental biochemical processes and the development of enzyme inhibitors as potential therapeutic agents. References 2023. Synthesis of pyrimidinone derivatives via Biginelli reaction using 2-nitrobenzaldehyde. Research on Chemical Intermediates, 49(12). DOI: 10.1007/s11164-023-05162-9 2019. Reduction of 2-nitrobenzaldehyde by chloramphenicol nitroreductase. Bioorganic & Medicinal Chemistry Letters, 29(10). DOI: 10.1016/j.bmcl.2019.02.025 2007. Detection of nitrofuran residues in honey using 2-nitrobenzaldehyde. Journal of Agricultural and Food Chemistry, 55(21). DOI: 10.1021/jf0625712 |
| Market Analysis Reports |