Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Changzhou Huaren Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huarenchem.com | |||
 | +86 (519) 8812-2965 | |||
 | +86 (519) 8810-4456 | |||
 | huarenchem@vip.163.com sales@huarenchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Qingdao Free Trade Zone United International Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.unitedint.com | |||
 | +86 (532) 83893696 +86 13808950921 | |||
 | +86 (532) 8389-3695 | |||
 | jason.wang@unitedint.com | |||
 | QQ Chat | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2006 | ||||
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| La Mesta Chimie Fine S.A.S. | France | |||
|---|---|---|---|---|
 | www.la-mesta.com | |||
 | +33 (4) 9208-5300 | |||
 | +33 (4) 9208-5340 | |||
 | info@la-mesta.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shijiazhuang Lida Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lid-ff.cn | |||
 | +86 (311) 8573-7996 | |||
 | +86 (311) 8573-9175 | |||
 | service@lid-ff.cn | |||
| Chemical manufacturer since 1993 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Dishman Pharmaceuticals & Chemicals Limited | USA | |||
|---|---|---|---|---|
 | www.dishmangroup.com | |||
 | +1 (732) 560-4300 | |||
 | +1 (732) 560-4343 | |||
 | bhaveshoza@dishmangroup.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Trignokem International | India | |||
|---|---|---|---|---|
 | www.trignokem.com | |||
 | +91 (40) 2717-0709 | |||
 | info@trignokem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Alder Research Chemicals Private Limited | India | |||
|---|---|---|---|---|
 | www.alderchemicals.com | |||
 | +91 (900) 301-4798 | |||
 | sales@alderchemicals.com | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Gansu Yunhao Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yunhaochem.cn | |||
 | +86 (0519) 8838-0792 | |||
 | 149443309@qq.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Zibo Fengbao Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fengbaochem.com | |||
 | +86 (533) 688-9529 688-9546 | |||
 | +86 (533) 688-5546 | |||
 | info@fengbaochem.com | |||
| Chemical manufacturer | ||||
| DeLong Chemicals America | USA | |||
|---|---|---|---|---|
 | www.delongchemicals.com | |||
 | +1 (203) 271-9017 | |||
 | sales@delongchemicals.com | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Taizhou Skyway Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.skywaychem.com | |||
 | +86 (576) 8547-1369 | |||
 | +86 (576) 8257-2096 | |||
 | skyway@skywaychem.com | |||
| Chemical manufacturer | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Classification | Flavors and spices >> Synthetic spice >> Lactone and oxygen-containing heterocyclic compound >> Thiazole, thiophene and pyridine |
|---|---|
| Name | 2-Acetylthiophene |
| Synonyms | 2-Acetyl thiophene; Methyl 2-thienyl ketone |
| Molecular Structure | 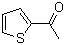 |
| Molecular Formula | C6H6OS |
| Molecular Weight | 126.17 |
| CAS Registry Number | 88-15-3 |
| EC Number | 201-804-6 |
| SMILES | CC(=O)C1=CC=CS1 |
| Density | 1.16 |
|---|---|
| Melting point | 10-11 °C |
| Boiling point | 214 °C |
| Refractive index | 1.564-1.568 |
| Flash point | 91 °C |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H311-H312-H332 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P262-P264-P270-P271-P280-P301+P317-P302+P352-P304+P340-P316-P317-P321-P330-P361+P364-P362+P364-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
2-Acetylthiophene is a sulfur-containing heterocyclic compound that was first synthesized in the early 20th century as part of the research on thiophene chemistry. These studies were aimed at understanding the reactivity and potential uses of thiophene derivatives, which have aromatic stability and versatile chemical properties. The compound consists of a thiophene ring that is substituted with an acetyl group at the second position, which imparts its unique chemical properties. Early studies focused on its synthesis and characterization, laying the foundation for its multiple applications in modern chemistry. The main application of 2-acetylthiophene is in the flavor and fragrance industry. Its unique aroma is reminiscent of roasted or caramelized foods, making it a valuable ingredient in creating flavor profiles in foods and spices. It provides nutty and roasted notes to products such as baked goods, confectionery, and salty snacks. In perfumes, it forms complex aroma components that add depth and warmth to perfume blends. In drug development, 2-acetylthiophene is used as an intermediate in the synthesis of a variety of biologically active compounds. Its structure allows the introduction of additional functional groups, making it a versatile building block in medicinal chemistry. It is particularly useful in creating compounds with potential antimicrobial, anti-inflammatory, and anticancer activities. The thiophene core can interact with biological targets, and the acetyl group can be further modified to enhance the pharmacological properties of the compound. The compound plays an important role in organic synthesis as a reagent for building more complex chemical entities. Its acetyl group is reactive and can undergo a variety of transformations, such as nucleophilic addition, condensation, and cyclization reactions. This versatility enables the synthesis of a wide range of heterocyclic compounds and other organic molecules, allowing the development of new synthetic methods and the exploration of new chemical space. In materials science, 2-acetylthiophene is used as a precursor for the synthesis of conjugated polymers and other advanced materials. Its thiophene ring contributes to the electronic properties of these materials, making them suitable for use in organic electronic devices such as organic photovoltaics (OPVs) and organic light-emitting diodes (OLEDs). The compound's ability to participate in polymerization reactions is valuable in the design of materials with tailored electrical and optical properties. The compound is also used to develop chemical sensors. Its unique structure and reactivity allow it to interact with a variety of analytes and can be used to create sensors for detecting chemicals in the environment, industry, and biomedicine. These sensors can be used to monitor pollutants, detect hazardous substances, and analyze biological samples, promoting the development of analytical chemistry. In research and development, 2-acetylthiophene is the subject of study to understand the properties and reactivity of thiophene derivatives. It is a model compound for studying the behavior of thiophene in chemical reactions, providing insights into its aromaticity, stability and functionalization potential. This research supports the discovery of new applications and the development of novel thiophene-based compounds. References 2023. RIFM fragrance ingredient safety assessment, 2-acetylthiophene, CAS registry number 88-15-3. Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 178. DOI: 10.1016/j.fct.2023.113704 2023. Synthesis, Characterization and Hirshfeld Surface Analysis of Some Thiosemicarbazones Containing a Five-Membered Ring. Journal of Structural Chemistry, 64(5). DOI: 10.1134/s0022476623050013 2023. Solvent-dependence of KI Mediated Electrosynthesis of Imidazo[1,2-a]pyridines. Chemical Research in Chinese Universities, 39(2). DOI: 10.1007/s40242-023-2323-y |
| Market Analysis Reports |