Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | 1,3,5-Tri(4-aminophenoxy)benzene |
| Molecular Structure | 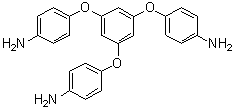 |
| Molecular Formula | C24H21N3O3 |
| Molecular Weight | 399.44 |
| CAS Registry Number | 102852-92-6 |
| EC Number | 878-922-5 |
| SMILES | C1=CC(=CC=C1N)OC2=CC(=CC(=C2)OC3=CC=C(C=C3)N)OC4=CC=C(C=C4)N |
| Solubility | Insoluble (3.7E-5 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.296±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 149-150 °C** |
| Boiling point | 618.7$+/-$55.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 356.0$+/-$25.2 $degree$C (Calc.)* |
| Index of refraction | 1.694 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2015 ACD/Labs) |
| ** | Abdalla, Mohamed A. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
1,3,5-Tri(4-aminophenoxy)benzene is a multifunctional aromatic compound consisting of a केंद्रीय benzene ring substituted at the 1,3,5-positions with three phenoxy groups, each bearing an amino group at the para position. Compounds of this type are widely studied and applied in polymer chemistry and materials science due to their symmetrical structure, multiple reactive amino groups, and ability to form highly crosslinked or branched polymer networks. The molecular structure features a central benzene core connected through ether linkages to three peripheral phenyl rings. Each of these outer rings carries an amino group at the 4-position, resulting in a total of three primary amine functionalities. The ether linkages provide flexibility compared to fully aromatic carbon–carbon connections, while still maintaining good thermal stability. The overall geometry is relatively rigid and symmetrical, which can influence the packing, solubility, and processing behavior of materials derived from this compound. The presence of multiple amino groups enables strong hydrogen bonding and high reactivity toward electrophilic reagents. The synthesis of 1,3,5-tri(4-aminophenoxy)benzene is typically achieved through nucleophilic aromatic substitution or Ullmann-type ether formation between a trihalogenated benzene core and protected aminophenol derivatives. After formation of the ether linkages, the amino groups may be introduced or deprotected through reduction of nitro precursors. These synthetic strategies allow precise placement of functional groups and preservation of the symmetrical structure, yielding a compound suitable for use as a multifunctional monomer. In polymer chemistry, this compound is widely used as a crosslinking agent or monomer in the preparation of high-performance polymers such as polyimides, polyamides, and epoxy resins. The three amino groups can react with dianhydrides, diacid chlorides, or epoxides to form highly crosslinked networks. These materials often exhibit excellent thermal stability, mechanical strength, and chemical resistance. The ether linkages in the structure can improve solubility and processability compared to more rigid aromatic systems, making the resulting polymers easier to fabricate into films or coatings. The compound is also important in the development of advanced materials with tailored properties. Its symmetrical, trifunctional structure allows for the design of network polymers with controlled crosslink density, which can influence properties such as glass transition temperature, elasticity, and dielectric behavior. Such materials are used in electronics, aerospace components, adhesives, and protective coatings. In addition to its role in polymer synthesis, 1,3,5-tri(4-aminophenoxy)benzene serves as a versatile intermediate in organic synthesis. The amino groups can undergo acylation, alkylation, or condensation reactions, enabling the preparation of a variety of derivatives. These derivatives can be used in the construction of functional molecules, including ligands, dyes, and other specialized materials. Overall, 1,3,5-tri(4-aminophenoxy)benzene is an important multifunctional aromatic compound characterized by a symmetrical structure, multiple amino groups, and ether linkages. Its combination of reactivity, stability, and structural features makes it a valuable building block in polymer chemistry, materials science, and advanced organic synthesis. References 2024. Advances in hyperbranched polymer chemistry. Iranian Polymer Journal. DOI: 10.1007/s13726-024-01379-6 2024. Lightweight and strong toughness of polyimide/SiO2 composite aerogels with super-high working temperature for high-performance thermal insulation and flame-resistant application. Journal of Materials Science. DOI: 10.1007/s10853-024-10087-8 2023. Development of polyimide aerogel stock shapes through polyimide aerogel particles. Journal of Porous Materials. DOI: 10.1007/s10934-023-01489-1 |
| Market Analysis Reports |