Online Database of Chemicals from Around the World
| Extrasynthese Chemical S.A.S. | France | |||
|---|---|---|---|---|
 | www.extrasynthese.com | |||
 | +33 (47) 898-2034 | |||
 | +33 (47) 898-1945 | |||
 | info@extrasynthese.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Aktin Chemicals Inc | China | |||
|---|---|---|---|---|
 | www.aktinchem.com | |||
 | +86 400-028-7725 | |||
 | +86 (28) 8515-2372 | |||
 | info@aktinchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Cfm Oskar Tropitzsch GmbH | Germany | |||
|---|---|---|---|---|
 | www.cfmot.de | |||
 | +49 (9231) 9619-0 | |||
 | +49 (9231) 9619-60 | |||
 | info@cfmot.de | |||
| Chemical distributor since 1985 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Naturewill Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.naturewillbio.com | |||
 | +86 (28) 8263-2533 | |||
 | info@naturewillbio.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Plant extracts |
|---|---|
| Name | Sarsasapogenin |
| Synonyms | Parigenin; (25S)-Spirostan-3b-ol; (25S)-5beta-Spirostan-3beta-ol |
| Molecular Structure | 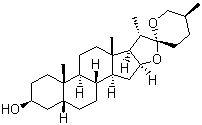 |
| Molecular Formula | C27H44O3 |
| Molecular Weight | 416.64 |
| CAS Registry Number | 126-19-2 |
| EC Number | 204-776-3 |
| SMILES | C[C@H]1CC[C@@]2([C@H]([C@H]3[C@@H](O2)C[C@@H]4[C@@]3(CC[C@H]5[C@H]4CC[C@H]6[C@@]5(CC[C@@H](C6)O)C)C)C)OC1 |
| Density | 1.1$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 200 - 201.5 $degree$C (Expl.) |
| Boiling point | 516.6$+/-$20.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 266.2$+/-$21.8 $degree$C (Calc.)* |
| Index of refraction | 1.552 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Sarsasapogenin is a naturally occurring steroidal sapogenin isolated primarily from plants of the genus Smilax and several other botanical sources. It is a spirostan-type steroidal compound that serves as an aglycone component of certain saponins. Structurally, it belongs to the class of steroidal sapogenins characterized by a tetracyclic steroid nucleus fused to a spiroketal side chain. The molecular structure of sarsasapogenin contains the characteristic cyclopentanoperhydrophenanthrene steroid framework composed of three six-membered rings and one five-membered ring. Attached to this steroid nucleus is a spiro-linked oxygen-containing ring system at the side chain, giving rise to the spirostan skeleton. The molecule also contains a hydroxyl group that contributes to its polarity and chemical reactivity. Sarsasapogenin occurs naturally as part of glycosidic saponins in plants. In these natural products, sugar chains are attached to the hydroxyl group of the sapogenin core. Hydrolysis of the glycosidic bonds yields free sarsasapogenin. Plants containing steroidal saponins have been studied extensively in phytochemistry because of their structural relationship to steroid hormones and their use as raw materials in steroid synthesis. Historically, steroidal sapogenins such as sarsasapogenin gained industrial importance in the mid-20th century as starting materials for the semisynthesis of corticosteroids, sex hormones, and other steroid pharmaceuticals. Their naturally occurring steroid framework provided a practical source of complex carbon skeletons before modern total synthesis methods became efficient. The stereochemistry of sarsasapogenin is highly defined and contributes significantly to its biological and physicochemical properties. The spiroketal side chain introduces conformational rigidity, while the multiple chiral centers create a three-dimensional structure similar to many endogenous steroids. In natural systems, saponins containing sarsasapogenin are believed to contribute to plant defense mechanisms against herbivores and microorganisms. The amphiphilic nature of saponins, arising from the combination of a hydrophobic steroid core and hydrophilic sugar groups, enables interaction with biological membranes. Sarsasapogenin itself has been investigated in pharmacological and biochemical research due to its structural similarity to steroid hormones and neuroactive steroids. Studies have explored its interactions with signaling pathways, lipid metabolism, and inflammatory processes. However, its primary established significance remains as a naturally occurring steroidal sapogenin and synthetic intermediate. From a synthetic chemistry perspective, sarsasapogenin serves as a valuable precursor in steroid modification reactions. The steroid nucleus and hydroxyl functionality allow introduction of additional substituents through oxidation, reduction, esterification, and side-chain transformation reactions. Such chemistry has historically been important in the preparation of steroid-derived pharmaceuticals. Physicochemically, sarsasapogenin is relatively hydrophobic due to its extensive fused-ring hydrocarbon framework, although the hydroxyl group and spiroketal oxygen atoms contribute some polarity. It is generally poorly soluble in water but soluble in many organic solvents. The discovery and characterization of sarsasapogenin contributed to the broader understanding of steroidal natural products and their relationship to biologically active steroids. Structural elucidation of sapogenins in the early development of natural products chemistry played an important role in advancing steroid chemistry and pharmaceutical synthesis. Overall, sarsasapogenin is a naturally occurring spirostan steroidal sapogenin found in certain plants as part of glycosidic saponins. Its rigid steroid framework and spiroketal side chain make it an important compound in phytochemistry and steroid synthesis, particularly as a natural precursor for semisynthetic steroidal compounds. References 2026. Investigation on the working mechanisms of sustainable corrosion inhibitors for mild steel in acid solutions: a review based on experimental and computational analysis. Surface Science and Technology. DOI: 10.1007/s44251-025-00119-z 2026. Diosgenin-mediated NF-κB and MAPK pathway modulation for osteoarthritis treatment: molecular mechanisms, bioavailability enhancement, and therapeutic applications. Inflammopharmacology. DOI: 10.1007/s10787-025-02102-4 |
| Market Analysis Reports |