Online Database of Chemicals from Around the World
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 2-(3-Aminophenyl)-1H-benzimidazol-6-amine |
| Synonyms | 2-(3-Aminophenyl)-1H-benzimidazol-6-amine; 5,3'-Diamino-2-phenylbenzimidazole |
| Molecular Structure | 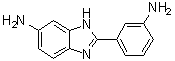 |
| Molecular Formula | C13H12N4 |
| Molecular Weight | 224.26 |
| CAS Registry Number | 13676-49-8 |
| EC Number | 988-207-0 |
| SMILES | C1=CC(=CC(=C1)N)C2=NC3=C(N2)C=C(C=C3)N |
| Solubility | Very slightly soluble (0.54 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.359±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 562.7$+/-$56.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 328.6$+/-$19.0 $degree$C (Calc.)* |
| Index of refraction | 1.787 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319 Details |
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details |
| SDS | Available |
|
2-(3-Aminophenyl)-1H-benzimidazol-6-amine is an aromatic heterocyclic compound consisting of a benzimidazole core substituted with amino groups at the 2-position on the phenyl ring and at the 6-position of the benzimidazole ring. Benzimidazole derivatives have been widely studied for their biological activity and utility as building blocks in medicinal chemistry and materials science. The presence of multiple amino groups in this compound increases its reactivity and potential for hydrogen bonding, making it a versatile intermediate in chemical synthesis. The molecular structure comprises a fused bicyclic system, where a benzene ring is fused to an imidazole ring, forming the benzimidazole core. The amino substituents introduce nucleophilic sites, enabling the compound to participate in a variety of reactions such as acylation, sulfonylation, and Schiff base formation. This reactivity is leveraged in the synthesis of functional molecules, including pharmaceuticals, ligands, and polymer precursors. The compound is typically synthesized through condensation reactions involving o-phenylenediamine derivatives and suitable carboxylic acid or aldehyde precursors. The positioning of amino groups is carefully controlled during synthesis to preserve the functionality of both the benzimidazole ring and the aromatic substituent. This ensures that the compound can serve as an effective intermediate for subsequent chemical transformations. In medicinal chemistry, 2-(3-aminophenyl)-1H-benzimidazol-6-amine and related derivatives have been investigated for their potential pharmacological activities, including antimicrobial, antiviral, and anticancer properties. The benzimidazole ring system is known to interact with biological targets through hydrogen bonding and pi-stacking interactions, while the amino groups enhance solubility and binding potential. Such structural features make it a valuable scaffold in drug design and discovery. The compound is also used as a precursor in the preparation of fluorescent dyes and ligands for coordination chemistry. Its amino groups can react with electrophilic compounds to form conjugates or functionalized derivatives suitable for imaging, sensing, and materials applications. In polymer chemistry, derivatives of this molecule can be incorporated into networks to introduce functional sites, improve thermal stability, or modify electronic properties. Overall, 2-(3-aminophenyl)-1H-benzimidazol-6-amine is a multifunctional heterocyclic compound with significant utility in organic synthesis, medicinal chemistry, and materials science. Its symmetrical arrangement of amino groups, reactivity, and benzimidazole core make it an important intermediate and building block for high-value applications. References 2019. Synthesis and properties of processable poly(benzimidazole-imide)s based on 2-(3-aminophenyl)-5-aminobenzimidazole. Journal of Polymer Research. DOI: 10.1007/s10965-019-1930-2 2002. Cyclization of 1-(Acylamino)- or 1-(Aroylamino)-2-aminoarenes. Science of Synthesis. |
| Market Analysis Reports |