Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Rhodia (shanghai) International Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rhodia.com | |||
 | +86 (21) 2408-9317 +86 13601753458 | |||
 | +86 (21) 5483-0035 | |||
 | hank.xue@ap.rhodia.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Euticals S.p.A. | Italy | |||
|---|---|---|---|---|
 | www.euticals.com | |||
 | +39 (371) 490-2339 | |||
 | +39 (371) 417-635 | |||
 | contact@archimica.com | |||
| Chemical manufacturer since 1946 | ||||
| Kemprotec Limited | UK | |||
|---|---|---|---|---|
 | www.kemprotec.com | |||
 | +44 (1642) 591-764 | |||
 | +44 (1845) 550-001 | |||
 | sales@kemprotec.co.uk | |||
| Chemical distributor since 1998 | ||||
| Archimica Inc. | USA | |||
|---|---|---|---|---|
 | www.archimica.com | |||
 | +1 (302) 994-3043 | |||
 | +1 (302) 994-3475 | |||
 | contact@archimica.com | |||
| Chemical manufacturer since 1946 | ||||
| Epsilon Chimie Chemical Manufacturer | France | |||
|---|---|---|---|---|
 | www.epsilon-chimie.com | |||
 | +33 (2) 9842-4650 | |||
 | +33 (2) 9842-4402 | |||
 | pierre.cornec@epsilon-chimie.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | Vinylphosphonic acid |
| Synonyms | ethenylphosphonic acid |
| Molecular Structure | 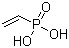 |
| Molecular Formula | C2H5O3P |
| Molecular Weight | 108.03 |
| CAS Registry Number | 1746-03-8 |
| EC Number | 217-123-2 |
| SMILES | C=CP(=O)(O)O |
| Density | 1.4$+/-$0.1 g/cm3 Calc.*, 1.37 g/mL (Expl.) |
|---|---|
| Melting point | 36 $degree$C (Expl.) |
| Boiling point | 266.2$+/-$23.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 114.8$+/-$22.6 $degree$C (Calc.)*, 113 $degree$C (Expl.) |
| Index of refraction | 1.466 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS05 Danger Details GHS05 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H290 Details | ||||||||||||||||||||
| Safety Statements | P280-P301+P330+P331-P303+P361+P353-P305+P351+P338 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 3265 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Vinylphosphonic acid is an organophosphorus compound containing both a vinyl group and a phosphonic acid functional group. Its molecular formula is C2H5O3P, and its structure can be represented as CH2=CH–P(=O)(OH)2. The molecule combines an unsaturated carbon–carbon double bond with a strongly acidic phosphonic acid moiety, giving it distinctive chemical and polymerization properties. Structurally, the compound consists of a terminal vinyl group (–CH=CH2) directly bonded to a phosphorus atom through a carbon–phosphorus bond. The phosphorus atom is tetrahedrally coordinated and bears one double-bonded oxygen atom and two hydroxyl groups. This phosphonic acid functionality differs from phosphoric acid esters because the phosphorus atom is directly bonded to carbon rather than oxygen. The phosphonic acid group is highly polar and strongly acidic. In aqueous solution, vinylphosphonic acid can undergo stepwise deprotonation to form monoanionic and dianionic phosphonate species depending on pH. These ionizable groups give the compound high water solubility and strong affinity for metal ions and mineral surfaces. The vinyl group is capable of free-radical polymerization, making vinylphosphonic acid an important functional monomer in polymer chemistry. During polymerization, the carbon–carbon double bond reacts to form polymer chains while the phosphonic acid groups remain pendant along the polymer backbone. These phosphonate-functionalized polymers exhibit high hydrophilicity, ion-binding capability, and proton conductivity. Because of its acidic phosphonate functionality, polymers derived from vinylphosphonic acid are widely investigated in ion-exchange materials, proton-conducting membranes, scale inhibitors, adhesives, coatings, and biomaterials. The phosphonic acid groups can strongly coordinate with metal ions such as calcium, magnesium, iron, and transition metals, which is important in surface modification and chelation applications. Chemically, the phosphonic acid group can undergo neutralization to form phosphonate salts, esterification to generate phosphonate esters, and condensation reactions under appropriate conditions. The strong P–C bond is chemically stable and resistant to hydrolysis compared with phosphate esters, which contributes to the durability of phosphonic acid derivatives. The vinyl functionality enables copolymerization with many other vinyl monomers such as acrylic acid, styrene, acrylamides, and vinyl acetate. This versatility allows tuning of polymer properties including hydrophilicity, charge density, adhesion, and thermal stability. Vinylphosphonic acid is commonly synthesized through phosphorus-containing precursor reactions involving vinyl halides, acetylene derivatives, or phosphite intermediates, followed by oxidation or hydrolysis steps to generate the phosphonic acid functionality. From a physicochemical perspective, the compound is highly polar and strongly hydrophilic due to the phosphonic acid group. In contrast, the vinyl portion contributes only limited hydrophobicity. The molecule readily forms hydrogen bonds and ionic interactions, influencing viscosity and intermolecular association in concentrated solutions. In materials science, phosphonic acid groups are valued because of their strong binding affinity to metal oxides and inorganic surfaces. Vinylphosphonic acid-containing polymers are therefore useful in coatings, corrosion inhibition, dental materials, and surface-functionalized nanoparticles. Historically, vinylphosphonic acid gained importance with the development of specialty functional polymers and ion-conducting materials. Interest increased further in areas such as fuel-cell membranes, biomedical polymers, and advanced coatings where stable phosphorus-containing functionality is advantageous. Overall, vinylphosphonic acid is a bifunctional organophosphorus compound containing a polymerizable vinyl group and a strongly acidic phosphonic acid moiety. Its unique combination of unsaturation, acidity, metal-binding capability, and polymer compatibility makes it an important monomer in functional polymer and materials chemistry. References 2025. Development of sustainable bio-based photocurable coatings on aluminum alloy surfaces. Journal of Coatings Technology and Research. DOI: 10.1007/s11998-025-01152-1 2025. Functional antifouling hydrogel surface engineering: insight into mechanisms, interfacial regulation and marine protection. Science China Materials. DOI: 10.1007/s40843-025-3479-9 2025. Use of Hemp (Cannabis sativa) Biomass Components and Activated Carbons Based on Them to Remove Pollutants from Aqueous Media: 1. Metal Ions. Polymer Science, Series D. DOI: 10.1134/s199542122570056x |
| Market Analysis Reports |