Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| Fluoropharm Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoropharm.com | |||
 | +86 (571) 8558-6753 +86 13336034509 | |||
 | +86 (571) 8558-6753 | |||
 | sales@fluoropharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Metropolitan Eximchem Ltd. | India | |||
|---|---|---|---|---|
 | www.metrosun.com | |||
 | +91 (22) 2408-1528 | |||
 | +91 (22) 2407-7375 | |||
 | info@metrosun.com | |||
| Chemical manufacturer since 1996 | ||||
| Classification | Organic raw materials >> Aryl compounds >> Anilines |
|---|---|
| Name | 2,5-Dichlorobenzene-1,4-diamine |
| Synonyms | 1,4-Diamino-2,5-dichlorobenzene |
| Molecular Structure | 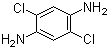 |
| Molecular Formula | C6H6Cl2N2 |
| Molecular Weight | 177.03 |
| CAS Registry Number | 20103-09-7 |
| EC Number | 243-512-1 |
| SMILES | C1=C(C(=CC(=C1Cl)N)Cl)N |
| Density | 1.5$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 164 - 168 $degree$C (Expl.) |
| Boiling point | 303.7$+/-$37.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 137.5$+/-$26.5 $degree$C (Calc.)* |
| Index of refraction | 1.679 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
2,5-Dichlorobenzene-1,4-diamine is an aromatic diamine compound characterized by two amino groups at the 1- and 4-positions of a benzene ring and two chlorine atoms at the 2- and 5-positions. This arrangement creates a symmetrical substitution pattern that combines nucleophilic amino functionalities with electron-withdrawing chloro substituents, influencing both the chemical reactivity and electronic properties of the molecule. It is primarily used as an intermediate in organic synthesis and polymer chemistry. The molecular structure consists of a benzene ring with para-oriented amino groups and ortho-positioned chlorine atoms relative to each amino group. The amino groups are highly reactive nucleophiles, capable of participating in acylation, sulfonylation, diazotization, and Schiff base formation. The chloro substituents are electron-withdrawing, which can reduce the basicity of the amino groups slightly and affect reaction selectivity. The combination of these functional groups allows the molecule to act as a building block for dyes, pharmaceuticals, and high-performance polymers. Synthesis of 2,5-dichlorobenzene-1,4-diamine typically involves chlorination of benzene derivatives followed by selective introduction of amino groups through nucleophilic substitution or reduction of nitro precursors. Protecting groups may be used to control the reactivity of the amino functionalities during multi-step syntheses. The resulting high-purity compound is suitable for downstream chemical transformations and industrial applications. In polymer chemistry, 2,5-dichlorobenzene-1,4-diamine is used as a monomer for the preparation of polyamides, polyimides, and epoxy resins. The diamine functionality enables the formation of linear or crosslinked polymer chains, while the rigid aromatic structure and chloro substituents impart thermal stability, mechanical strength, and chemical resistance. Such polymers find applications in coatings, adhesives, and electronic materials where durability and stability are essential. The compound also serves as an intermediate in the synthesis of aromatic dyes and pigments. Its amino groups can undergo diazotization and subsequent coupling reactions to form azo dyes with specific color properties. The chloro substituents can further modify the chromophore properties or improve the solubility and processability of the final dye. In addition, 2,5-dichlorobenzene-1,4-diamine has applications in medicinal and pharmaceutical chemistry, where its functionalized aromatic structure can be used to construct heterocyclic scaffolds or bioactive molecules. Its reactive amino sites allow the introduction of additional substituents or conjugation to other pharmacophores, making it a versatile intermediate for drug design. Overall, 2,5-dichlorobenzene-1,4-diamine is a multifunctional aromatic diamine with significant utility in polymer synthesis, dye chemistry, and organic synthesis. Its combination of nucleophilic amino groups, electron-withdrawing chloro substituents, and rigid aromatic framework make it an important building block for high-performance materials and functional compounds. References 2022. Investigation of Synthesis, Characterization, and Biological Activities of Water-Soluble Polymer with Horseradish Peroxidase Enzyme. Polymer Science, Series B. DOI: 10.1134/s1560090422700488 2022. Synthesis of Organosoluble Polyimides and Optical Fiber Protective Coatings on Their Basis. Polymer Science, Series B. DOI: 10.1134/s1560090420010078 2017. Synthesis and properties of chloro-containing polyimides. Polymer Science, Series B. DOI: 10.1134/s1560090417040091 |
| Market Analysis Reports |