Online Database of Chemicals from Around the World
| CAS: 20638-07-7 Product: 9,9-Bis(3-amino-4-hydroxyphenyl)fluorene No suppliers available. |
| Classification | Organic raw materials >> Heterocyclic compound >> Fluorenes |
|---|---|
| Name | 9,9-Bis(3-amino-4-hydroxyphenyl)fluorene |
| Synonyms | 2-amino-4-[9-(3-amino-4-hydroxyphenyl)fluoren-9-yl]phenol |
| Molecular Structure | 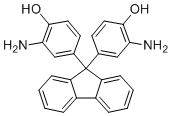 |
| Molecular Formula | C25H20N2O2 |
| Molecular Weight | 380.44 |
| CAS Registry Number | 20638-07-7 |
| EC Number | 678-172-7 |
| SMILES | C1=CC=C2C(=C1)C3=CC=CC=C3C2(C4=CC(=C(C=C4)O)N)C5=CC(=C(C=C5)O)N |
| Density | 1.4$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 596.7$+/-$50.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 314.7$+/-$30.1 $degree$C (Calc.)* |
| Index of refraction | 1.769 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
9,9-Bis(3-amino-4-hydroxyphenyl)fluorene is a multifunctional aromatic compound based on a fluorene core substituted at the 9-position with two phenyl rings, each bearing amino and hydroxyl groups. Compounds of this type are important in polymer chemistry, materials science, and advanced organic synthesis due to their rigid structure, multiple reactive functional groups, and ability to form extended conjugated systems. The presence of both amino and hydroxyl substituents enables diverse chemical transformations, including condensation, polymerization, and crosslinking reactions. The molecular structure consists of a fluorene backbone, a tricyclic aromatic system in which two benzene rings are fused to a central five-membered ring. At the 9-position, two substituted phenyl groups are attached, each containing an amino group at the 3-position and a hydroxyl group at the 4-position. This arrangement creates a symmetrical molecule with multiple hydrogen-bonding sites and nucleophilic centers. The rigid fluorene core imparts thermal stability and limits rotational freedom, which is beneficial for maintaining defined molecular geometry in polymers and functional materials. The synthesis of 9,9-bis(3-amino-4-hydroxyphenyl)fluorene typically involves the condensation of fluorene derivatives with substituted phenols followed by nitration and reduction steps to introduce the amino groups. Controlled reaction conditions are required to ensure substitution at the correct positions and to avoid overreaction or degradation of the aromatic system. The resulting compound is generally obtained as a solid with good thermal stability and can be purified for use in further chemical transformations. In polymer chemistry, this compound is widely used as a monomer or intermediate for the preparation of high-performance polymers such as polyimides, polyamides, and epoxy resins. The amino groups can react with dianhydrides, diacids, or epoxides to form polymer chains, while the hydroxyl groups can participate in additional crosslinking or modification reactions. The fluorene core contributes rigidity and thermal resistance, while the substituted phenyl groups enhance solubility and processability of the resulting polymers. These properties make such materials suitable for applications in electronics, coatings, and structural components. In addition to polymer applications, 9,9-bis(3-amino-4-hydroxyphenyl)fluorene is used in the development of functional materials, including optical and electronic devices. The extended conjugated system allows for light absorption and emission, making fluorene derivatives useful in organic light-emitting diodes and related technologies. The presence of multiple functional groups enables further modification to tailor optical, electronic, or mechanical properties for specific applications. The compound also serves as a versatile intermediate in organic synthesis. The amino groups can undergo acylation, alkylation, or condensation, while the hydroxyl groups can be etherified or esterified. This versatility allows the construction of complex molecular architectures and the generation of derivatives with controlled reactivity and physical properties. The combination of rigidity, multiple functional sites, and aromatic character makes it a valuable scaffold for designing advanced materials and chemical systems. Overall, 9,9-bis(3-amino-4-hydroxyphenyl)fluorene is an important multifunctional aromatic compound that integrates a rigid fluorene core with reactive amino and hydroxyl substituents. Its structural features provide thermal stability, chemical versatility, and suitability for polymer synthesis and material applications, making it a key intermediate in modern materials chemistry and organic synthesis. References 2007. Comparative study of the bimolecular electron transfer of fullerenes (C60/C70) and 9,9-disubstituted fluorenes by laser flash photolysis. Photochemical & photobiological sciences : Official journal of the European Photochemistry Association and the European Society for Photobiology. DOI: 10.1039/b617814f |
| Market Analysis Reports |