Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Junkai (Tianjin) Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.junkaichem.com | |||
 | +86 (22) 8568-9515 ex 8818 +86 13920864700 | |||
 | +86 (22) 2378-7875 | |||
 | sales@junkaichem.com jane-jkchem@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2007 | ||||
| CHINATECH (TIANJIN) CHEMICAL CO., LTD. | China | |||
|---|---|---|---|---|
 | www.chinatechem.com | |||
 | +86 (22) 8372-6202 EXT8819 | |||
 | sales4@chinatechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Jinzhou Zhongshi Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jzzhongshi.com | |||
 | +86 (311) 8448-7556 | |||
 | +86 (311) 8448-7368 | |||
 | zhongshichemical@163.com | |||
| Chemical manufacturer since 2005 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | 1,4-Bis(4-aminophenoxy)benzene |
| Synonyms | 4-[4-(4-aminophenoxy)phenoxy]aniline |
| Molecular Structure | 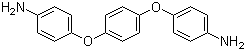 |
| Molecular Formula | C18H16N2O2 |
| Molecular Weight | 292.33 |
| CAS Registry Number | 3491-12-1 |
| EC Number | 677-976-5 |
| SMILES | InChI=1S/C18H16N2O2/c19-13-1-5-15(6-2-13)21-17-9-11-18(12-10-17)22-16-7-3-14(20)4-8-16 |
| Density | 1.2$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 173 $degree$C (Expl.) |
| Boiling point | 483.6$+/-$40.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 267.8$+/-$21.0 $degree$C (Calc.)* |
| Index of refraction | 1.668 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335-H400 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
1,4-Bis(4-aminophenoxy)benzene is an aromatic diamine compound in which two 4-aminophenoxy groups are attached to the para positions of a central benzene ring. The molecule features a linear arrangement of aromatic rings connected via ether linkages, with amino groups positioned para to the oxygen atoms. This combination of nucleophilic amino groups, ether connectivity, and rigid aromatic framework makes the compound a valuable intermediate in polymer synthesis, organic synthesis, and high-performance material development. The molecular structure consists of a central benzene ring substituted at the 1- and 4-positions with oxygen atoms linking to 4-aminophenyl groups. The para-positioned amino groups provide reactive sites capable of participating in acylation, sulfonylation, and polycondensation reactions. The ether linkages contribute flexibility to the molecule, while the aromatic rings enhance thermal stability and rigidity, which are important properties for polymers and high-performance materials derived from this compound. Synthesis of 1,4-bis(4-aminophenoxy)benzene generally involves nucleophilic aromatic substitution of 1,4-dihalobenzenes with 4-nitrophenol, followed by reduction of the nitro groups to form the diamine. Reaction conditions are carefully controlled to achieve complete conversion while minimizing side reactions, ensuring high purity of the diamine. The resulting compound is suitable for use as a monomer in the preparation of polymers and as an intermediate for functional aromatic derivatives. A major application of this compound is in the production of polyimides, polyamides, and epoxy resins. The diamine functionality enables polymer chain extension and crosslinking, while the rigid aromatic backbone and ether linkages provide thermal stability, mechanical strength, and chemical resistance. Polymers derived from 1,4-bis(4-aminophenoxy)benzene are used in advanced coatings, adhesives, electronic materials, and composite applications where durability and performance at high temperatures are required. In addition to polymer applications, 1,4-bis(4-aminophenoxy)benzene serves as a building block in organic synthesis. Its amino groups can undergo derivatization, including formation of Schiff bases, amides, and sulfonamides, allowing chemists to prepare functionalized molecules for material science, pharmaceutical intermediates, and ligands for coordination chemistry. The combination of reactive amino sites and a structurally rigid yet flexible framework makes it a versatile intermediate for chemical synthesis. Overall, 1,4-bis(4-aminophenoxy)benzene is a multifunctional aromatic diamine with significant utility in high-performance polymer synthesis, organic chemistry, and material science. Its combination of para-oriented amino groups, ether linkages, and rigid aromatic structure provides both reactivity and stability, making it an important intermediate for the design and production of advanced materials and functional chemical compounds. References 2025. Advanced strategies for thermal conductivity enhancement in polymer composites for application as thermal management materials. Macromolecular Research. DOI: 10.1007/s13233-025-00377-8 2024. High-frequency low-dielectric-loss in linear-backbone-structured polyimides with ester groups and ether bonds. Communications Materials. DOI: 10.1038/s43246-024-00502-7 2023. Research progress of filled-type high-thermal-conductivity flexible polyimide composites: a review. Journal of Materials Science. DOI: 10.1007/s10853-023-09040-y |
| Market Analysis Reports |