Online Database of Chemicals from Around the World
| Cymtek Laboratories, Ltd. | China | |||
|---|---|---|---|---|
 | www.cymteklabs.com | |||
 | +86 (519) 8109-0216 | |||
 | chemservice@outlook.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Amine |
|---|---|
| Name | 3-(4-Aminophenyl)-2,3-dihydro-1,1,3-trimethyl-1H-inden-5-amine |
| Synonyms | 5-Amino-3-(4-aminophenyl)-1,1,3-trimethylindan |
| Molecular Structure | 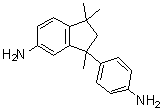 |
| Molecular Formula | C18H22N2 |
| Molecular Weight | 266.38 |
| CAS Registry Number | 54628-90-9 |
| EC Number | 611-176-9 |
| SMILES | CC1(CC(C2=C1C=CC(=C2)N)(C)C3=CC=C(C=C3)N)C |
| Solubility | Practically insoluble (0.013 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.091±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 93-94 °C (heptane )** |
| Boiling point | 432.3$+/-$45.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 257.9$+/-$28.2 $degree$C (Calc.)* |
| Index of refraction | 1.616 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2017 ACD/Labs) |
| ** | Petropoulos, John C. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
|
3-(4-Aminophenyl)-2,3-dihydro-1,1,3-trimethyl-1H-inden-5-amine is a polyfunctional aromatic amine compound featuring a substituted indane core bonded to a 4-aminophenyl group and bearing multiple methyl and amino substituents. The structure integrates a rigid bicyclic framework with both primary amino groups and sterically demanding methyl groups, providing a combination of chemical reactivity, structural rigidity, and steric effects. These properties make the compound valuable as an intermediate in organic synthesis, polymer chemistry, and the development of functional aromatic derivatives. The molecular structure consists of a 2,3-dihydro-1H-indene ring substituted with three methyl groups at positions 1 and 3 and an amino group at the 5-position. A 4-aminophenyl group is attached at the 3-position of the indane ring, providing an additional nucleophilic site. The two amino groups enable reactions such as acylation, sulfonylation, Schiff base formation, and condensation reactions, while the methyl groups influence steric accessibility and electronic distribution across the molecule. The combination of the bicyclic indane framework and planar phenyl substituent contributes to the rigidity and overall molecular shape, which can affect reactivity and intermolecular interactions. Synthesis of 3-(4-aminophenyl)-2,3-dihydro-1,1,3-trimethyl-1H-inden-5-amine generally involves functionalization of an appropriately substituted indanone or indene precursor, followed by the introduction of amino groups through reduction of nitro intermediates or direct amination. Reaction conditions are carefully optimized to maintain regioselectivity, control stereochemistry, and prevent overreaction, ensuring high purity and yield of the diamine product. Protective groups may be employed during multi-step syntheses to selectively manipulate one amino group while leaving the other intact. This compound finds applications primarily as an intermediate in the synthesis of dyes, pigments, and specialty polymers. The combination of the amino-functionalized phenyl group and indane core allows it to participate in the formation of azo or azomethine dyes with defined color properties. In polymer chemistry, the diamine functionality enables crosslinking and polymer chain extension, while the rigid indane framework imparts thermal stability and mechanical strength to derived polymers. Additionally, 3-(4-aminophenyl)-2,3-dihydro-1,1,3-trimethyl-1H-inden-5-amine is utilized in organic synthesis for constructing heterocyclic compounds, functionalized aromatic systems, and other bioactive molecules. The multiple reactive sites, combined with steric and electronic effects from the methyl substituents, allow chemists to design molecules with controlled reactivity, enhanced stability, or targeted functional properties. Overall, 3-(4-aminophenyl)-2,3-dihydro-1,1,3-trimethyl-1H-inden-5-amine is a multifunctional aromatic diamine with versatile applications in organic synthesis, dye chemistry, and high-performance polymer development. Its combination of nucleophilic amino groups, sterically significant methyl substituents, and rigid indane framework provides both chemical reactivity and structural stability, making it an important intermediate for advanced materials and functional compounds. References 2024. Liquid crystal alignment agent for photo-alignment, liquid crystal alignment film and liquid crystal display element. TW-I865340-B. URL: https://patents.google.com/patent/TWI865340B/en 2023. Liquid crystal alignment agent, liquid crystal alignment film, and liquid crystal display element. WO-2025127092-A1. URL: https://patents.google.com/patent/WO2025127092A1/en 2023. Liquid crystal aligning agent, liquid crystal alignment film and liquid crystal display element. WO-2025121249-A1. URL: https://patents.google.com/patent/WO2025121249A1/en |
| Market Analysis Reports |