Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tyger Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.tygersci.com | |||
 | +1 (609) 434-0144 | |||
 | +1 (609) 434-0143 | |||
 | sales@tygersci.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Winchem Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-chemical.com | |||
 | +86 (574) 8385-1061 | |||
 | +86 (574) 8708-3209 | |||
 | info@win-chemical.com hhp13@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Wuhan Comings Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | comings.en.made-in-china.com | |||
 | +86 13659820406 | |||
 | info@comingschemi.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Metropolitan Eximchem Ltd. | India | |||
|---|---|---|---|---|
 | www.metrosun.com | |||
 | +91 (22) 2408-1528 | |||
 | +91 (22) 2407-7375 | |||
 | info@metrosun.com | |||
| Chemical manufacturer since 1996 | ||||
| Classification | Chemical reagent >> Organic reagent >> Polyamine |
|---|---|
| Name | 2-Chloro-1,4-diaminobenzene |
| Synonyms | 2-Chloro-p-phenylenediamine |
| Molecular Structure | 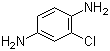 |
| Molecular Formula | C6H7ClN2 |
| Molecular Weight | 142.59 |
| CAS Registry Number | 615-66-7 |
| EC Number | 210-441-2 |
| SMILES | C1=CC(=C(C=C1N)Cl)N |
| Density | 1.3$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 62-66 $degree$C (Expl.) |
| Boiling point | 287.9$+/-$20.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 127.9$+/-$21.8 $degree$C (Calc.)* |
| Index of refraction | 1.671 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312+H332-H302-H312-H315-H319-H332 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P321-P330-P332+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-Chloro-1,4-diaminobenzene is an aromatic diamine compound in which two amino groups are attached to the 1- and 4-positions of a benzene ring, and a chlorine atom is substituted at the 2-position. This substitution pattern combines the nucleophilic reactivity of the amino groups with the electron-withdrawing effect of the chloro substituent, making the molecule useful as an intermediate in organic synthesis, dye chemistry, and polymer production. The presence of the chlorine atom can influence both the chemical reactivity and the physical properties of the compound, such as solubility and thermal stability. The molecular structure consists of a benzene ring with amino groups at the para positions (1 and 4) and a chlorine atom at the ortho position relative to one of the amino groups. The amino groups serve as nucleophilic centers that can participate in acylation, sulfonylation, diazotization, and polycondensation reactions. The chlorine atom, being electron-withdrawing, slightly deactivates the aromatic ring toward electrophilic substitution, which can be exploited to direct reactions selectively. Synthesis of 2-chloro-1,4-diaminobenzene typically involves selective chlorination of 1,4-diaminobenzene (para-phenylenediamine) under controlled conditions to ensure the chlorine atom is introduced at the 2-position without over-chlorination or oxidation of the amino groups. Alternative methods may start from chloronitrobenzene derivatives followed by reduction of nitro groups to yield the diamine. Reaction parameters such as temperature, solvent, and stoichiometry are carefully managed to achieve high-purity product suitable for downstream applications. One of the primary applications of 2-chloro-1,4-diaminobenzene is as an intermediate in the synthesis of dyes and pigments. The amino groups can undergo diazotization followed by coupling with phenols, aromatic amines, or other nucleophiles to form azo dyes with defined color properties. The chloro substituent can influence hue, stability, and solubility of the resulting dyes, making it an important design element in color chemistry. In polymer chemistry, this diamine is used as a monomer for high-performance polymers such as polyamides, polyimides, and epoxy curing agents. The para-oriented amino groups enable chain extension or crosslinking, while the ortho chlorine can influence the polymer backbone’s rigidity, solubility, and thermal properties. Polymers derived from 2-chloro-1,4-diaminobenzene are employed in coatings, adhesives, and composite materials where chemical resistance and durability are required. Additionally, 2-chloro-1,4-diaminobenzene serves as a versatile intermediate in organic synthesis for producing heterocyclic compounds, functionalized aromatic molecules, and other chemical derivatives. Its combination of reactive amino groups and a modulating chloro substituent allows for selective chemical transformations and the development of materials with tailored properties. Overall, 2-chloro-1,4-diaminobenzene is a multifunctional aromatic diamine with important applications in dye chemistry, polymer synthesis, and organic synthesis. Its para-oriented amino groups, ortho chloro substituent, and rigid aromatic framework provide both chemical reactivity and structural stability, making it a valuable intermediate for functional materials and advanced chemical compounds. References 2020. Gas-Transport Properties of Polyimides with Various Side Groups. Polymer Science, Series C. DOI: 10.1134/s1811238220020058 2020. Synthesis of Organosoluble Polyimides and Optical Fiber Protective Coatings on Their Basis. Polymer Science, Series B. DOI: 10.1134/s1560090420010078 2017. Synthesis and properties of chloro-containing polyimides. Polymer Science, Series B. DOI: 10.1134/s1560090417040091 |
| Market Analysis Reports |