Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Changzhou Sunchem Pharmaceutical Chemical Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunkechem.com | |||
 | +86 (519) 8519-5575 8519-5528 8519-5565 | |||
 | +86 (519) 8519-5585 | |||
 | info@sunkechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Synasia Inc | USA | |||
|---|---|---|---|---|
 | www.synasia.com | |||
 | +1 (732) 205-9880 | |||
 | +1 (732) 205-1788 | |||
 | info@synasia.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Topfine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.topfine-chem.com | |||
 | +86 (21) 5858-0600 | |||
 | +86 (21) 5858-6316 | |||
 | gavin@topfine-chem.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Wuhan Comings Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | comings.en.made-in-china.com | |||
 | +86 13659820406 | |||
 | info@comingschemi.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Shanghai Shenqiang Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shenqiang.com | |||
 | +86 (21) 6297-1188 6297-1788 | |||
 | +86 (21) 6257-8811 | |||
 | shenqiang@shenqiang.com | |||
| Chemical manufacturer since 1996 | ||||
| Classification | Organic raw materials >> Aryl compounds >> Anilines |
|---|---|
| Name | 4,4'-Diaminobenzanilide |
| Molecular Structure | 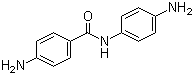 |
| Molecular Formula | C13H13N3O |
| Molecular Weight | 227.26 |
| Protein Sequence | X |
| CAS Registry Number | 785-30-8 |
| EC Number | 212-321-5 |
| SMILES | C1=CC(=CC=C1C(=O)NC2=CC=C(C=C2)N)N |
| Density | 1.3$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 205 - 207 $degree$C (Expl.) |
| Boiling point | 384.0$+/-$27.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 186.0$+/-$23.7 $degree$C (Calc.)* |
| Index of refraction | 1.734 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
4,4'-Diaminobenzanilide is an aromatic diamine compound consisting of two benzene rings linked by an amide (–CONH–) group, with each ring substituted by a primary amino group at the para (4-) position relative to the amide linkage. This structural arrangement combines nucleophilic amino groups with a polar amide bond, providing both chemical reactivity and the potential for hydrogen bonding. The compound is primarily used as an intermediate in polymer synthesis, organic chemistry, and the production of high-performance materials. The molecular structure features a central amide linkage connecting two benzene rings, each bearing a para-amino group. The amino groups can participate in condensation and crosslinking reactions, while the amide group imparts rigidity and polarity to the molecule. This combination of functional groups allows for controlled polymerization reactions and the formation of thermally stable polymers. The planar nature of the amide bond also contributes to intermolecular hydrogen bonding, affecting solubility and crystallinity in derived materials. Synthesis of 4,4'-diaminobenzanilide typically involves the acylation of 4-nitroaniline derivatives with 4-aminobenzoyl chloride, followed by reduction of the nitro groups to amino groups. Careful control of reaction conditions is required to prevent over-acylation or undesired side reactions. The resulting diamine is generally obtained as a solid with good purity, suitable for subsequent polymerization or chemical modification. One of the main applications of 4,4'-diaminobenzanilide is as a monomer or intermediate in the production of high-performance polymers, including polyamides, polyimides, and epoxy resins. Its para-amino groups enable chain extension and crosslinking, while the amide linkage provides thermal and chemical stability. Polymers derived from this compound are used in adhesives, coatings, electronic materials, and composite materials where mechanical strength and resistance to heat and chemicals are important. In addition to polymer applications, 4,4'-diaminobenzanilide serves as a building block for dyes, pigments, and functionalized aromatic compounds. Its amino and amide groups allow selective chemical transformations, enabling the synthesis of molecules with tailored properties for industrial or research applications. Overall, 4,4'-diaminobenzanilide is a versatile aromatic diamine with an amide linkage that provides chemical reactivity, structural rigidity, and thermal stability. Its combination of nucleophilic sites and polar linkage makes it a valuable intermediate in high-performance polymers, functional materials, and specialty chemical synthesis. References 2024. Effect of Low-temperature Imidization on Properties and Aggregation Structures of Polyimide Films with Different Rigidity. Chinese Journal of Polymer Science. DOI: 10.1007/s10118-024-3137-1 2023. Preparation and Characterization of New Soluble and Thermally Stable Polyazomethine by Polycondensation of Thiophene-2,5-dicarboxaldehyde and Ortho-tolidine for Optoelectronics. Polymer Science, Series B. DOI: 10.1134/s1560090423701038 2023. Influence of diamine moieties on the gas permeation performances of polyimide: perspectives from experiment and simulation. Journal of Polymer Research. DOI: 10.1007/s10965-023-03677-8 |
| Market Analysis Reports |