Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Zhejiang Kangfeng Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kangfeng.com | |||
 | +86 (579) 8670-9298 8670-9155 | |||
 | +86 (579) 8670-9678 | |||
 | wukang@kangfeng.com qimaoxin@kangfeng.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Changzhou Sunchem Pharmaceutical Chemical Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunkechem.com | |||
 | +86 (519) 8519-5575 8519-5528 8519-5565 | |||
 | +86 (519) 8519-5585 | |||
 | info@sunkechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Future Industrial Limited By Share Ltd. | China | |||
|---|---|---|---|---|
 | www.jonln.com | |||
 | +86 400-0066-400 | |||
 | +86 (21) 5566-0885 | |||
 | sales@jonln.com | |||
| Chemical distributor since 2011 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Topfine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.topfine-chem.com | |||
 | +86 (21) 5858-0600 | |||
 | +86 (21) 5858-6316 | |||
 | gavin@topfine-chem.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| EAST STAR BIOTECH (SUZHOU) CO., LTD. | China | |||
|---|---|---|---|---|
 | www.eaststarbio.com | |||
 | +86 17706846989 | |||
 | daniel.creations007@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Jiangsu Fenghua Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fhchem.com | |||
 | +86 (515) 8855-1818 | |||
 | +86 (515) 8856-2028 | |||
 | sales@fhchem.com | |||
| Chemical manufacturer | ||||
| Marshallton Research Laboratories | USA | |||
|---|---|---|---|---|
 | www.marshalltonlabs.com | |||
 | +1 (336) 983-2131 | |||
 | +1 (336) 983-0096 | |||
 | marshallton@windstream.net | |||
| Chemical manufacturer since 1974 | ||||
| Aaron Chemistry GmbH | Germany | |||
|---|---|---|---|---|
 | www.aaron-chemistry.de | |||
 | +49 (8823) 917-521 | |||
 | +49 (8823) 917-523 | |||
 | sales@aaron-chemistry.de | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Dongyang Dongnong Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dongnongchem.com | |||
 | +86 (579) 8667-5549 8667-5569 | |||
 | +86 (579) 8667-5483 | |||
 | zjh@dongnongchem.com | |||
| Chemical manufacturer | ||||
| Xinyi Chemical Research Institute | China | |||
|---|---|---|---|---|
 | www.xinyichemical.com | |||
 | +86 (519) 8646-2478 8647-1022 | |||
 | +86 (519) 8646-8478 | |||
 | lxy.mq@163.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Dalian Honkai Chemical Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.honkai.com | |||
 | +86 (411) 8479-1969 | |||
 | +86 (411) 8482-0706 | |||
 | sales@honkai.com | |||
| Chemical manufacturer since 2005 | ||||
| Frinton Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.frinton.com | |||
 | +1 (856) 722-7037 | |||
 | +1 (856) 439-1977 | |||
 | sales@frinton.com frinton@frinton.com | |||
| Chemical manufacturer since 1963 | ||||
| Classification | Organic raw materials >> Alcohols, phenols, phenolic compounds and derivatives |
|---|---|
| Name | 3-Trifluoromethylphenol |
| Synonyms | m-Hydroxybenzotrifluoride; 3-Hydroxybenzotrifluoride; 3-(Trifluoromethyl)phenol; alpha,alpha,alpha-Trifluoro-m-cresol |
| Molecular Structure | 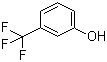 |
| Molecular Formula | C7H5F3O |
| Molecular Weight | 162.11 |
| CAS Registry Number | 98-17-9 |
| EC Number | 202-645-5 |
| SMILES | C1=CC(=CC(=C1)O)C(F)(F)F |
| Density | 1.3$+/-$0.1 g/cm3 Calc.*, 1.333 g/mL (Expl.) |
|---|---|
| Melting point | -2 - -1.8 $degree$C (Expl.) |
| Boiling point | 178.0 $degree$C 760 mmHg (Calc.)*, 178 - 179 $degree$C (Expl.) |
| Flash point | 73.9 $degree$C (Calc.)*, 73 $degree$C (Expl.) |
| Index of refraction | 1.458 (Calc.)*, 1.458 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS06;GHS07 Danger Details GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H302-H312-H314-H315-H318-H319-H335-H412 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P271-P273-P280-P301+P316-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P351+P338-P305+P354+P338-P316-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
3-Trifluoromethylphenol is an aromatic phenolic compound containing both a hydroxyl group and a trifluoromethyl substituent attached to a benzene ring. Its molecular formula is C7H5F3O. The structure consists of a phenol core in which a trifluoromethyl group (–CF3) is located at the 3-position (meta position) relative to the hydroxyl group. The molecule combines two chemically significant functional groups: a phenolic hydroxyl group and a strongly electron-withdrawing trifluoromethyl substituent. The hydroxyl group imparts weak acidity, hydrogen-bonding capability, and nucleophilic behavior through the oxygen atom, while the –CF3 group strongly affects the electronic properties of the aromatic ring. The trifluoromethyl group is one of the most powerful electron-withdrawing substituents used in organic chemistry. Through its strong inductive effect, it decreases electron density in the aromatic ring and influences both reactivity and acidity. In 3-trifluoromethylphenol, this effect increases the acidity of the phenolic hydroxyl group compared with unsubstituted phenol, because the conjugate phenoxide ion is stabilized by the electron-withdrawing substituent. The phenolic hydroxyl group can undergo typical phenol reactions, including esterification, etherification, oxidation, and deprotonation under basic conditions to form phenoxide salts. The aromatic ring may also participate in electrophilic substitution reactions, although the strongly deactivating –CF3 group reduces overall ring reactivity compared with simple phenols. The meta relationship between the hydroxyl and trifluoromethyl groups affects the directing effects during aromatic substitution. The hydroxyl group is an activating ortho/para director, whereas the trifluoromethyl group is a deactivating meta director. The combined influence of these substituents determines regioselectivity in synthetic transformations. Fluorinated phenols such as 3-trifluoromethylphenol are important intermediates in medicinal chemistry, agrochemical synthesis, and specialty materials chemistry. The introduction of fluorine-containing groups often improves metabolic stability, lipophilicity, oxidative resistance, and biological activity in target molecules. The trifluoromethyl substituent also increases hydrophobicity while maintaining a relatively compact steric profile. This balance of hydrophobic and electronic effects makes CF3-substituted aromatic compounds especially valuable in pharmaceutical design. Synthetically, 3-trifluoromethylphenol can be prepared through several routes, including hydroxylation of trifluoromethyl-substituted aromatic precursors, selective trifluoromethylation of phenol derivatives, or diazotization–hydrolysis sequences starting from corresponding amino compounds. From a physicochemical perspective, the molecule exhibits moderate polarity due to the hydroxyl group, while the aromatic ring and trifluoromethyl group contribute hydrophobic character. The strong carbon–fluorine bonds confer high thermal and chemical stability. The hydroxyl group allows intermolecular hydrogen bonding, which influences boiling point and solubility. However, the presence of the hydrophobic CF3 group limits water solubility relative to simple phenol. Spectroscopically, the compound displays characteristic aromatic and phenolic ultraviolet absorption features, while the fluorine atoms strongly influence nuclear magnetic resonance behavior due to fluorine coupling effects. Historically, trifluoromethyl-substituted aromatic compounds gained major industrial importance with the expansion of organofluorine chemistry in pharmaceuticals and agrochemicals during the 20th century. Their unique combination of electronic and physicochemical properties made them valuable building blocks in modern synthetic chemistry. Overall, 3-trifluoromethylphenol is a meta-substituted fluorinated phenol characterized by a hydroxyl group and a strongly electron-withdrawing trifluoromethyl substituent on an aromatic ring. Its altered acidity, enhanced chemical stability, and fluorinated aromatic character make it an important intermediate in organic, medicinal, and materials chemistry. References 2025. Development of novel fluoro-substituted rivastigmine derivatives as selective AChE inhibitors for the treatment of AD. Medicinal Chemistry Research. DOI: 10.1007/s00044-024-03250-y 2022. Functionalizing aromatic compounds with optical cycling centres. Nature Chemistry. DOI: 10.1038/s41557-022-00998-x |
| Market Analysis Reports |